Hemoglobin and Iron Metabolism

On this page
🩸 The Oxygen Express: Hemoglobin's Molecular Mastery
Hemoglobin stands as one of medicine's most elegant molecular machines, binding and releasing oxygen through cooperative precision while revealing disease through predictable clinical signatures. You'll master how its quaternary structure drives allosteric behavior, recognize the distinct patterns of anemia and hemoglobinopathies, and apply evidence-based strategies to diagnose and manage disorders from iron deficiency to sickle cell crisis. By integrating molecular mechanisms with physiological networks and clinical reasoning, you'll transform hemoglobin from a lab value into a diagnostic and therapeutic command center that guides patient care across specialties.
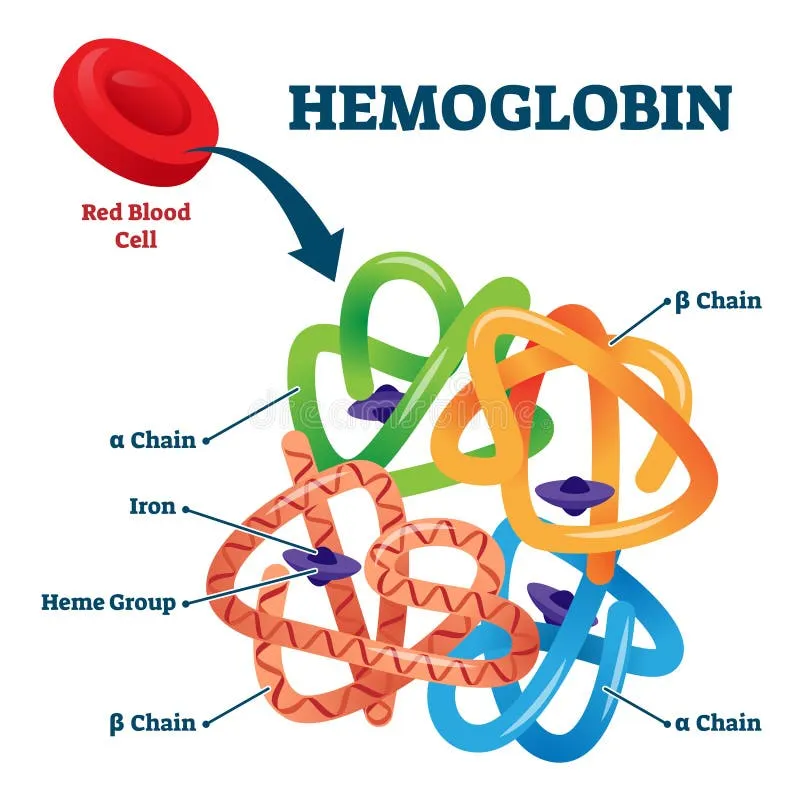
Hemoglobin's Quaternary Architecture
Hemoglobin represents one of medicine's most elegant molecular machines, consisting of 4 globin chains (2α, 2β) each cradling a heme group containing ferrous iron (Fe²⁺). This tetrameric structure enables cooperative oxygen binding, where each successive oxygen molecule binds with increasing affinity - a phenomenon creating the characteristic sigmoid oxygen-dissociation curve.
- Adult Hemoglobin (HbA): α₂β₂ - comprises 96-98% of normal adult hemoglobin
- α-chains: 141 amino acids each, encoded on chromosome 16
- β-chains: 146 amino acids each, encoded on chromosome 11
- Normal concentration: 12-16 g/dL (women), 14-18 g/dL (men)
- Fetal Hemoglobin (HbF): α₂γ₂ - <2% in normal adults
- Higher oxygen affinity than HbA (P₅₀ = 19 mmHg vs 27 mmHg)
- Resistant to acid elution and alkali denaturation
- Minor Hemoglobin (HbA₂): α₂δ₂ - 2-3% of normal adult hemoglobin
- Elevated in β-thalassemia trait (>3.5%)
📌 Remember: CAGE for globin chain locations - Chromosome 16 has Alpha chains, Gamma chains; chromosome 11 has Everything else (β, δ, ε chains)
Iron's Central Command in Heme Groups
Each heme group contains protoporphyrin IX complexed with ferrous iron (Fe²⁺) in the center, creating the oxygen-binding site. The iron must remain in the Fe²⁺ state for oxygen transport - oxidation to Fe³⁺ creates methemoglobin, which cannot carry oxygen.
| Parameter | Normal Value | Clinical Significance | Pathological Range |
|---|---|---|---|
| Heme Iron State | Fe²⁺ (ferrous) | Oxygen binding capable | Fe³⁺ = methemoglobin |
| Methemoglobin % | <1.5% | Normal oxidation | >10% = cyanosis |
| Oxygen Capacity | 1.34 mL O₂/g Hb | Theoretical maximum | Reduced in CO poisoning |
| Heme Groups/Hb | 4 per molecule | Cooperative binding | Mutation affects all 4 |
| Iron Content | 3.4 mg/g Hb | Iron requirement | Depleted in IDA |
The proximal histidine (F8) directly coordinates with iron, while the distal histidine (E7) stabilizes oxygen binding and prevents irreversible oxidation. This precise geometric arrangement allows reversible oxygen binding while preventing carbon monoxide from binding 250 times more readily than oxygen.
💡 Master This: Iron in the Fe³⁺ state shifts the oxygen-dissociation curve dramatically leftward with P₅₀ approaching zero - explaining why methemoglobinemia causes functional anemia despite normal hemoglobin levels
Understanding this molecular foundation predicts how genetic mutations, oxidative stress, and iron metabolism disorders disrupt oxygen transport, setting the stage for mastering hemoglobin's dynamic regulatory mechanisms.
🩸 The Oxygen Express: Hemoglobin's Molecular Mastery
⚙️ The Cooperative Engine: Hemoglobin's Allosteric Mastery
The T-State to R-State Transformation
Hemoglobin exists in two primary conformational states that determine oxygen affinity through quaternary structural changes:
- T-State (Tense): Low oxygen affinity conformation
- Deoxyhemoglobin predominant state
- Salt bridges between globin chains create rigid structure
- P₅₀ = 26-27 mmHg in this state
- Favored by 2,3-BPG binding in central cavity
- R-State (Relaxed): High oxygen affinity conformation
- Oxyhemoglobin predominant state
- Broken salt bridges allow flexible structure
- 10-fold higher oxygen affinity than T-state
- 2,3-BPG cannot bind effectively
📌 Remember: TENSE = Tight binding of 2,3-BPG, Easy No oxygen, Salt bridges Everywhere
Allosteric Effectors: The Molecular Rheostat
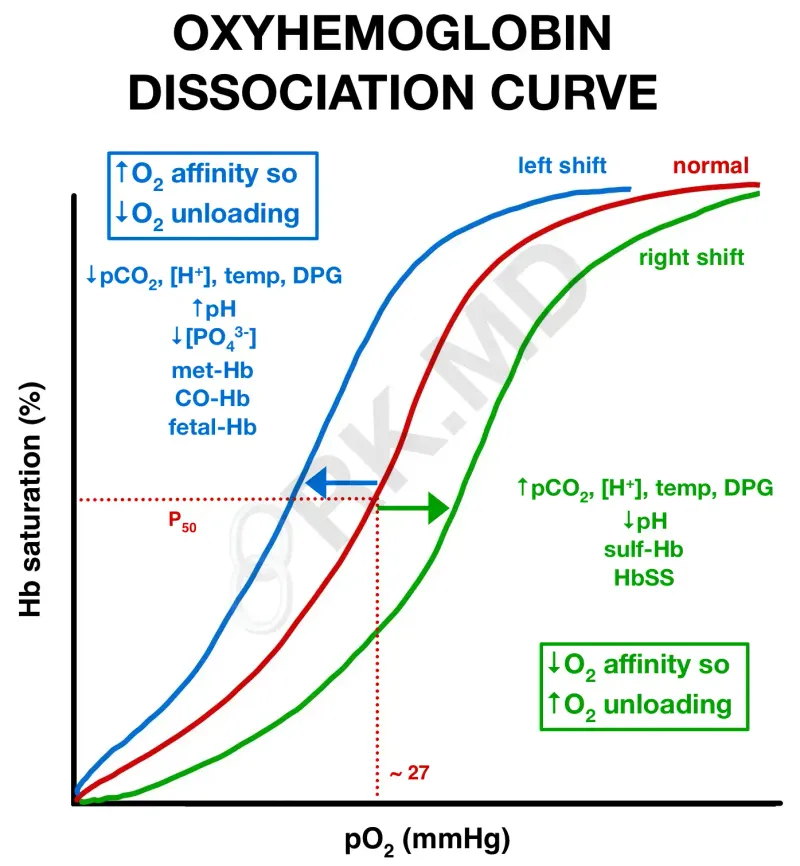
Hemoglobin's oxygen affinity responds to 4 primary allosteric effectors that shift the oxygen-dissociation curve based on metabolic demands:
| Effector | Mechanism | Curve Shift | Clinical Impact |
|---|---|---|---|
| 2,3-BPG | Binds T-state cavity | Right shift | ↑ O₂ unloading |
| H⁺ (pH) | Protonates histidines | Right shift | Bohr effect |
| CO₂ | Forms carbamino compounds | Right shift | Enhanced unloading |
| Temperature | Increases molecular motion | Right shift | Fever response |
| CO | Irreversible heme binding | Left shift | Toxic hypoxia |
The Bohr effect demonstrates hemoglobin's pH sensitivity, where decreased pH (increased H⁺) promotes oxygen release in metabolically active tissues producing CO₂ and lactic acid. This creates a physiological coupling between tissue metabolism and oxygen delivery.
💡 Master This: Root effect in fish hemoglobin shows complete oxygen loss at low pH, while human hemoglobin shows graded response - explaining why severe acidosis (pH <7.1) dramatically impairs oxygen transport despite normal hemoglobin levels
Cooperative Binding Mathematics
The Hill coefficient (n) quantifies cooperative binding, where n = 1 indicates no cooperation (myoglobin) and n = 2.8 represents normal hemoglobin cooperation:
$$\text{Fractional Saturation} = \frac{(PO_2)^n}{(P_{50})^n + (PO_2)^n}$$
This mathematical relationship explains why hemoglobin achieves 98% saturation at PO₂ = 100 mmHg (lungs) but drops to 75% saturation at PO₂ = 40 mmHg (tissues), creating the steep portion of the sigmoid curve that optimizes oxygen extraction.
Understanding these allosteric mechanisms reveals how hemoglobinopathies, metabolic disorders, and environmental factors alter oxygen transport efficiency, preparing you to analyze complex clinical presentations involving oxygen delivery dysfunction.
⚙️ The Cooperative Engine: Hemoglobin's Allosteric Mastery
🎯 Pattern Recognition: Hemoglobin's Clinical Signatures
The "See This, Think That" Framework
Systematic pattern recognition transforms complex hemoglobin disorders into predictable diagnostic pathways:
- Chocolate-Brown Blood + Normal PO₂ + Low O₂ Sat
- Think: Methemoglobinemia
- Key: Blood doesn't turn red with oxygen exposure
- Threshold: >10% methemoglobin causes visible cyanosis
- Mechanism: Fe³⁺ cannot bind oxygen
- Cherry-Red Blood + High O₂ Sat + Tissue Hypoxia
- Think: Carbon monoxide poisoning
- Key: COHb >20% causes symptoms
- Mechanism: CO affinity 250× > oxygen
- Trap: Pulse oximetry reads falsely normal
- Low Hb + Microcytic + Right-Shifted Curve
- Think: Iron deficiency anemia
- Key: Decreased 2,3-BPG initially, then increased as compensation
- P₅₀: Initially <25 mmHg, then >30 mmHg
- Normal Hb + Left-Shifted Curve + Family History
- Think: High-affinity hemoglobin variant
- Key: P₅₀ <20 mmHg with secondary polycythemia
- Mechanism: Impaired oxygen release stimulates EPO
📌 Remember: CHOCOLATE for methemoglobinemia - Chocolate blood, High methemoglobin, Oxygen can't bind, Cyanosis present, Oxidizing agents, Low functional Hb, Antidotes work, Treatment urgent, Emergency condition
Hemoglobin Electrophoresis Patterns
| Hemoglobin Type | Normal % | Migration Pattern | Clinical Significance |
|---|---|---|---|
| HbA (α₂β₂) | 96-98% | Standard mobility | Normal adult Hb |
| HbA₂ (α₂δ₂) | 2-3% | Slower than HbA | ↑ in β-thalassemia trait |
| HbF (α₂γ₂) | <2% | Alkali resistant | ↑ in β-thalassemia major |
| HbS (α₂β₆ᴳˡᵘ→ⱽᵃˡ) | 0% | Slower migration | Sickle cell disease |
| HbC (α₂β₆ᴳˡᵘ→ᴸʸˢ) | 0% | Slowest migration | Mild hemolytic anemia |
Oxygen-Dissociation Curve Shift Patterns
-
Left-Shifted Curves (P₅₀ <25 mmHg):
- High-affinity variants: P₅₀ = 12-20 mmHg
- Methemoglobinemia: Functional left shift of remaining normal Hb
- Carbon monoxide: COHb doesn't release oxygen
- Clinical effect: Tissue hypoxia despite normal/high O₂ saturation
-
Right-Shifted Curves (P₅₀ >30 mmHg):
- Chronic hypoxia: 2,3-BPG increases 50-100%
- Acidosis: pH <7.35 shifts curve right
- Hyperthermia: Fever >38.5°C decreases affinity
- Clinical effect: Enhanced oxygen unloading but impaired loading
💡 Master This: P₅₀ values predict clinical outcomes - P₅₀ <20 mmHg causes tissue hypoxia and secondary polycythemia, while P₅₀ >35 mmHg causes exercise intolerance and compensatory tachycardia
These pattern recognition frameworks enable rapid differentiation between structural variants, quantitative defects, and functional abnormalities, guiding targeted diagnostic workups and therapeutic interventions based on specific hemoglobin pathophysiology.
🎯 Pattern Recognition: Hemoglobin's Clinical Signatures
🔬 Diagnostic Discrimination: Unraveling Hemoglobin Mysteries
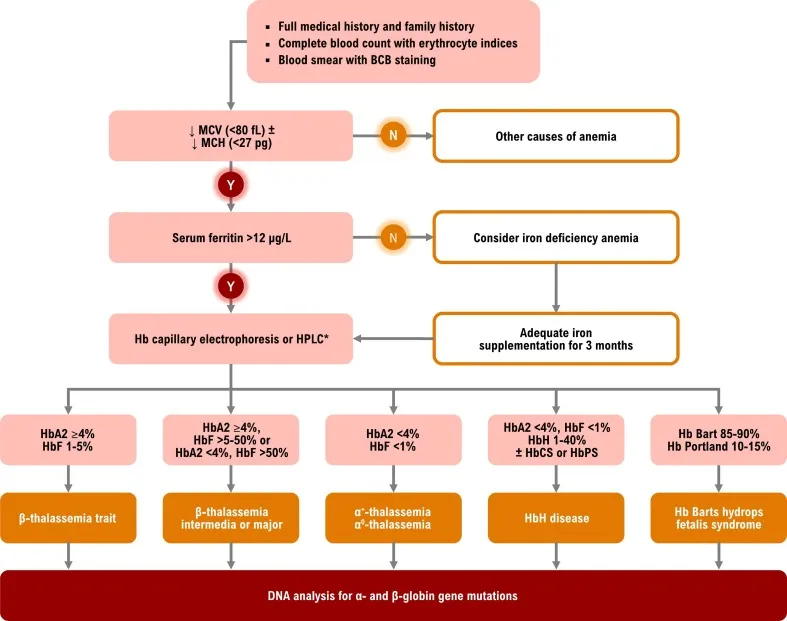
Quantitative vs. Qualitative Defect Discrimination
The fundamental distinction between thalassemias (reduced globin chain production) and hemoglobinopathies (abnormal globin chain structure) creates predictable laboratory patterns:
| Parameter | β-Thalassemia | Sickle Cell Disease | Iron Deficiency | Normal |
|---|---|---|---|---|
| Hb Level | 9-11 g/dL | 6-9 g/dL | 4-8 g/dL | 12-16 g/dL |
| MCV | 55-70 fL | 80-95 fL | 50-70 fL | 80-100 fL |
| RBC Count | ↑↑ (>5.5M) | Normal/↓ | ↓↓ | 4.5-5.5M |
| HbA₂ | ↑ (>3.5%) | Normal (2-3%) | ↓ (<2%) | 2-3% |
| HbF | ↑ (2-10%) | ↑ (5-15%) | Normal (<2%) | <2% |
| Iron Studies | Normal | Normal | ↓↓ Ferritin | Normal |
| Electrophoresis | Normal pattern | HbS band | Normal | Normal |
Functional Hemoglobin Assessment Matrix
- Oxygen Saturation Gap Analysis:
- Calculated O₂ Sat = (PO₂ × 0.003 + Hb × 1.34 × %Sat) ÷ (Hb × 1.34) × 100
- Measured O₂ Sat via co-oximetry (not pulse oximetry)
- Gap >5% suggests dyshemoglobin (MetHb, COHb)
- Normal gap with symptoms suggests high-affinity variant
⭐ Clinical Pearl: Pulse oximetry cannot distinguish COHb or MetHb from normal HbO₂ - always use co-oximetry when oxygen saturation gap exists or cyanosis occurs with normal PO₂
Advanced Discriminatory Testing
| Test | Indication | Key Findings | Clinical Threshold |
|---|---|---|---|
| P₅₀ Measurement | Suspected affinity variant | Normal: 26-28 mmHg | <20 or >32 mmHg abnormal |
| Kleihauer-Betke | HbF quantification | Acid-resistant cells | >2% adults abnormal |
| HPLC Analysis | Precise Hb quantification | Separates all variants | HbA₂ >3.5% = β-thal trait |
| DNA Analysis | Definitive diagnosis | Specific mutations | Gold standard confirmation |
| Osmotic Fragility | Membrane stability | Spherocyte detection | Increased in hereditary spherocytosis |
- Heat stability: HbH precipitates at 50°C in 1 hour
- Isopropanol test: Unstable variants precipitate in 20 minutes
- Acid elution: HbF resists 0.25M NaOH for 2 minutes
💡 Master This: Iron deficiency can mask β-thalassemia trait by normalizing HbA₂ levels and reducing MCV further - always recheck HbA₂ after iron repletion when microcytosis persists despite normal iron studies
Clinical Context Integration
The Mentzer Index provides rapid discrimination between iron deficiency and thalassemia trait:
$$\text{Mentzer Index} = \frac{\text{MCV}}{\text{RBC Count}}$$
- Mentzer Index <13: Suggests thalassemia trait (85% sensitivity)
- Mentzer Index >13: Suggests iron deficiency (97% specificity)
- Index 11-15: Overlap zone requiring additional testing
England-Fraser Index offers alternative discrimination: $$\text{E-F Index} = \text{MCV} - \text{RBC} - (5 × \text{Hb}) - 3.4$$
- Negative value: Thalassemia trait likely
- Positive value: Iron deficiency likely
These discriminatory tools enable systematic differentiation between complex hemoglobin disorders, guiding appropriate therapeutic interventions and genetic counseling based on precise pathophysiological mechanisms rather than empirical treatment approaches.
🔬 Diagnostic Discrimination: Unraveling Hemoglobin Mysteries
🎯 Therapeutic Precision: Evidence-Based Hemoglobin Management
Iron Deficiency Anemia: Precision Repletion Protocol
Oral Iron Optimization:
- Ferrous sulfate: 325mg TID = 195mg elemental iron daily
- Absorption enhancement: Vitamin C 500mg with each dose
- Timing: 1 hour before meals or 2 hours after for maximum absorption
- Expected response: Hb rise 1-2 g/dL in 2-4 weeks
- Duration: Continue 3-6 months after Hb normalization to replete stores
| Iron Preparation | Elemental Iron | Dosing | Absorption Rate |
|---|---|---|---|
| Ferrous Sulfate | 65mg/325mg tab | 325mg TID | 10-15% absorbed |
| Ferrous Gluconate | 38mg/325mg tab | 325mg TID | 12-18% absorbed |
| Ferrous Fumarate | 106mg/325mg tab | 200mg TID | 8-12% absorbed |
| Polysaccharide Iron | 150mg/capsule | 150mg BID | 15-20% absorbed |
Sickle Cell Disease: Comprehensive Crisis Prevention
Hydroxyurea Therapy Protocol:
- Starting dose: 15mg/kg/day (maximum 35mg/kg/day)
- Target: HbF >20% and 50% reduction in crisis frequency
- Monitoring: CBC weekly for 8 weeks, then monthly
- Dose escalation: Increase by 5mg/kg every 8 weeks if tolerated
- Hold criteria: ANC <2000, Platelets <80,000, Hb <4.5 g/dL
| Parameter | Baseline | Target | Monitoring Frequency |
|---|---|---|---|
| HbF % | 2-15% | >20% | Every 3 months |
| Crisis Rate | Variable | 50% reduction | Continuous tracking |
| ANC | >2500 | >2000 | Weekly × 8, then monthly |
| Platelets | >150,000 | >80,000 | Weekly × 8, then monthly |
| MCV | 80-95 fL | >100 fL | Monthly |
Methemoglobinemia: Emergency Antidote Protocols
Methylene Blue Administration:
- Indication: MetHb >20% or symptomatic at >10%
- Dose: 1-2 mg/kg IV over 5 minutes (maximum 7 mg/kg/day)
- Mechanism: NADPH-dependent reduction of MetHb to HbO₂
- Response: MetHb reduction within 30-60 minutes
- Contraindication: G6PD deficiency (can cause hemolysis)
💡 Master This: Methylene blue is contraindicated in G6PD deficiency because it depletes NADPH, causing severe hemolysis - use exchange transfusion or hyperbaric oxygen instead
Transfusion Thresholds and Targets
| Clinical Scenario | Hb Threshold | Target Hb | Special Considerations |
|---|---|---|---|
| Asymptomatic IDA | <7 g/dL | 8-10 g/dL | Avoid over-transfusion |
| Cardiac Disease | <8 g/dL | 9-11 g/dL | Higher threshold needed |
| Sickle Cell Crisis | <5 g/dL | 9-11 g/dL | Avoid >11 g/dL (hyperviscosity) |
| Acute MetHb | Any level | Normal Hb | Emergency antidote priority |
| Thalassemia Major | <9 g/dL | 9.5-10.5 g/dL | Prevent iron overload |
- Deferasirox: 20-40 mg/kg/day orally
- Deferoxamine: 20-40 mg/kg subcutaneous 8-12 hours daily
- Target ferritin: 500-1000 ng/mL
- Cardiac monitoring: Annual echo and T2 MRI*
These evidence-based protocols optimize therapeutic outcomes while minimizing complications, transforming hemoglobin disorders from life-limiting conditions into manageable chronic diseases through precision medicine approaches.
🎯 Therapeutic Precision: Evidence-Based Hemoglobin Management
🌐 Systems Integration: Hemoglobin's Physiological Networks
The Erythropoietin-Iron-Hemoglobin Axis
Erythropoietin (EPO) serves as the master regulator of red cell production, responding to tissue hypoxia through renal oxygen sensors and creating feedback loops that integrate iron availability, hemoglobin synthesis, and oxygen delivery:
- EPO Response Kinetics:
- Normal EPO: 5-30 mU/mL
- Hypoxia response: 10-fold increase within 24-48 hours
- Peak levels: 100-1000 mU/mL in severe anemia
- Half-life: 6-8 hours (requires continuous production)
| Condition | EPO Level | Iron Status | Hb Response | Mechanism |
|---|---|---|---|---|
| Iron Deficiency | ↑↑ (>100) | ↓ Stores | Poor | Substrate limitation |
| Chronic Disease | ↑ (30-100) | ↓ Available | Poor | Hepcidin block |
| Renal Disease | ↓ (<30) | Normal | Poor | EPO deficiency |
| Hemolysis | ↑↑ (>200) | ↑ Available | Good | Increased turnover |
Cardiovascular-Hemoglobin Integration
The cardiovascular system adapts to hemoglobin abnormalities through compensatory mechanisms that maintain tissue oxygen delivery but can lead to cardiac complications over time:
-
Acute Anemia Adaptations (Hb <8 g/dL):
- Cardiac output increases 30-50%
- Heart rate increases 20-40 bpm
- Stroke volume increases 15-25%
- Systemic vascular resistance decreases 20-30%
- 2,3-BPG increases 50-100% (right-shift curve)
-
Chronic Anemia Complications:
- High-output heart failure when Hb <6 g/dL chronically
- Left ventricular hypertrophy develops over months
- Pulmonary hypertension in sickle cell disease
- Iron cardiomyopathy in transfusion-dependent patients
📌 Remember: CARDIAC compensation in anemia - Cardiac output up, Afterload down, Rate increases, Delivery maintained, Increased stroke volume, Adaptation limits, Complications develop
Respiratory-Hemoglobin Coupling
Pulmonary function and hemoglobin create bidirectional interactions where lung disease affects oxygen loading and hemoglobin disorders influence respiratory drive:
| Parameter | Normal | Anemia Response | Hypoxic Lung Disease |
|---|---|---|---|
| Respiratory Rate | 12-20/min | ↑ 15-25% | ↑ 30-50% |
| Tidal Volume | 500 mL | ↑ 10-20% | ↑ 20-40% |
| Minute Ventilation | 6-8 L/min | ↑ 25-40% | ↑ 50-100% |
| P₅₀ | 27 mmHg | ↑ 30-32 mmHg | ↑ 28-35 mmHg |
| 2,3-BPG | 4-5 mmol/L | ↑ 6-8 mmol/L | ↑ 7-10 mmol/L |
Acid-Base-Hemoglobin Interactions
Hemoglobin serves as a major blood buffer (contributing 75% of blood buffering capacity) while acid-base status significantly affects oxygen affinity:
$$\text{Hb Buffer Capacity} = 2.3 \times \text{Hb concentration (g/dL)}$$
- Bohr Effect Quantification:
- ΔpH = 0.1 causes ΔP₅₀ = 1.2 mmHg
- Acidosis (pH 7.2): P₅₀ = 32 mmHg (enhanced unloading)
- Alkalosis (pH 7.6): P₅₀ = 22 mmHg (impaired unloading)
These integrated networks explain why complex patients with multiple comorbidities require systems-based approaches rather than isolated hemoglobin correction, setting the foundation for comprehensive clinical mastery of oxygen transport disorders.
🌐 Systems Integration: Hemoglobin's Physiological Networks
⚡ Clinical Command Center: Hemoglobin Mastery Arsenal
Essential Clinical Thresholds
| Parameter | Normal Range | Action Threshold | Emergency Level | Intervention |
|---|---|---|---|---|
| Hemoglobin | 12-16 g/dL (F), 14-18 g/dL (M) | <10 g/dL | <7 g/dL | Transfusion consideration |
| Methemoglobin | <1.5% | >10% | >20% | Methylene blue |
| COHb | <2% | >10% | >25% | Hyperbaric oxygen |
| P₅₀ | 26-28 mmHg | <20 or >32 mmHg | <15 or >40 mmHg | Variant workup |
| HbA₂ | 2-3% | >3.5% | N/A | β-thalassemia trait |
| Reticulocytes | 0.5-2% | <0.5% or >5% | <0.2% or >15% | Bone marrow evaluation |
Rapid Diagnostic Decision Tree
High-Yield Clinical Pearls
⭐ Emergency Pearl: Chocolate-brown blood that doesn't turn red with oxygen exposure = methemoglobinemia - treat with methylene blue 1-2 mg/kg IV if >20% or symptomatic >10%
⭐ Diagnostic Pearl: Microcytic anemia with normal iron studies + HbA₂ >3.5% = β-thalassemia trait - genetic counseling required, no iron supplementation
⭐ Therapeutic Pearl: Sickle cell crisis + Hb >11 g/dL = avoid transfusion (hyperviscosity risk) - use exchange transfusion if Hb >13 g/dL needed
💡 Master This: Pulse oximetry reads falsely normal in CO poisoning and methemoglobinemia - always use co-oximetry when clinical hypoxia exists with normal pulse ox
Rapid Reference Formulas
Oxygen Content Calculation: $$\text{O}_2 \text{ Content} = (1.34 \times \text{Hb} \times \text{SaO}_2) + (0.003 \times \text{PaO}_2)$$
Mentzer Index (Thalassemia vs Iron Deficiency): $$\text{Mentzer Index} = \frac{\text{MCV}}{\text{RBC Count}}$$
- <13: Thalassemia likely
- >13: Iron deficiency likely
Iron Deficit Calculation: $$\text{Iron Deficit (mg)} = \text{Weight (kg)} \times (15 - \text{Current Hb}) \times 2.4 + 500$$
Emergency Protocols
| Emergency | Recognition | Immediate Action | Definitive Treatment |
|---|---|---|---|
| Severe Anemia | Hb <7 + symptoms | Type & crossmatch | Packed RBCs |
| MetHb Crisis | Cyanosis + brown blood | ABG with co-ox | Methylene blue |
| CO Poisoning | Cherry red + normal PO₂ | 100% O₂ | Hyperbaric chamber |
| Sickle Crisis | Pain + sickle history | IV fluids + analgesia | Hydroxyurea/exchange |
| Acute Hemolysis | Dark urine + anemia | LDH, haptoglobin | Treat underlying cause |
This clinical arsenal enables rapid, accurate diagnosis and evidence-based management of hemoglobin disorders, transforming complex pathophysiology into systematic clinical excellence that optimizes patient outcomes through precision medicine approaches.
⚡ Clinical Command Center: Hemoglobin Mastery Arsenal
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























