Free Radicals: Basics - Unstable Atoms Alert
- Atoms or molecules with one or more unpaired electrons in their outer orbital; highly unstable and reactive.
- Seek stability by oxidizing (stealing electrons from) or reducing (donating electrons to) other molecules.
- Key Types:
- Reactive Oxygen Species (ROS):
- Superoxide ($O_2^{\cdot-}$)
- Hydroxyl radical ($\cdot OH$) - extremely reactive.
- Hydrogen peroxide ($H_2O_2$) (not a radical, but a key ROS)
- Reactive Nitrogen Species (RNS):
- Nitric oxide ($NO\cdot$)
- Peroxynitrite ($ONOO^-$)
- Reactive Oxygen Species (ROS):
⭐ The hydroxyl radical ($\cdot OH$) is the most reactive free radical, causing significant damage to DNA, lipids, and proteins. It has a very short half-life (nanoseconds).
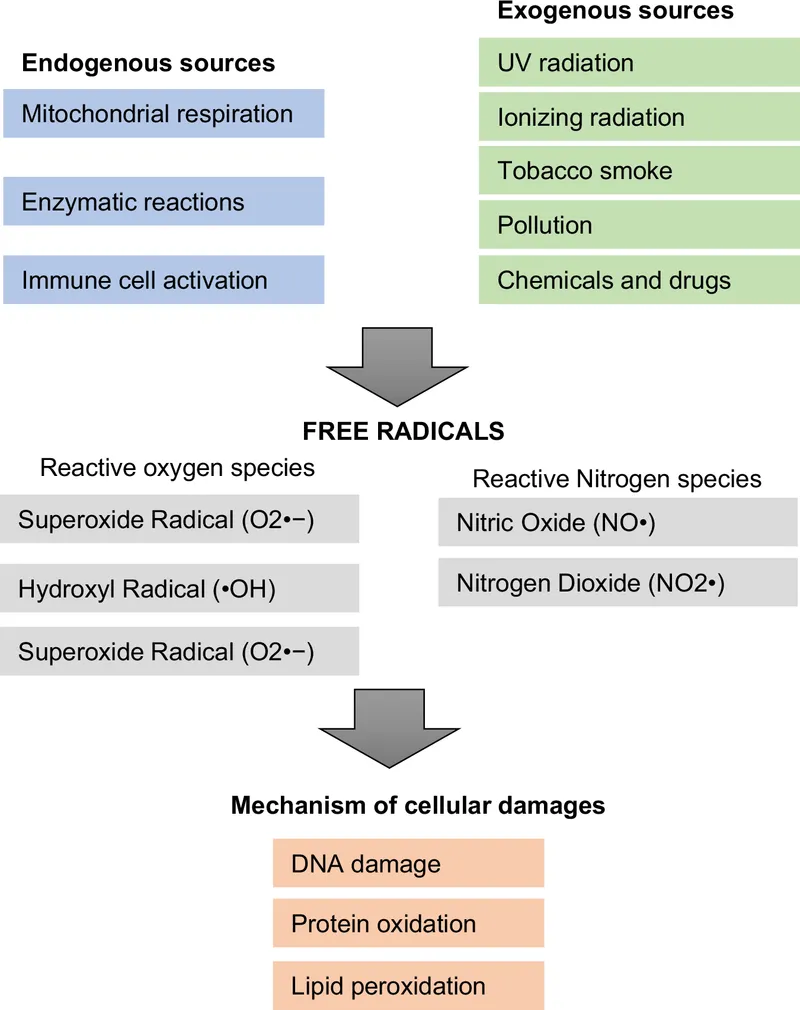
FR Generation - Where Mischief Brews

- Free Radicals (FRs): Unstable molecules, unpaired electrons, cause cellular damage.
- Endogenous Sources (Cellular Metabolism):
- Mitochondrial ETC: Major site; 'leaks' superoxide ($O_2^{\cdot-}$).
- NADPH Oxidase: Phagocytes for microbial killing (respiratory burst).
- Xanthine Oxidase: Purine degradation to uric acid.
- Fenton Reaction: $Fe^{2+} + H_2O_2 \rightarrow Fe^{3+} + \cdot OH + OH^-$. Iron/copper catalyzes hydroxyl radical ($\cdot OH$) formation.
- Peroxisomal metabolism: e.g., fatty acid oxidation.
- Exogenous Sources (Environmental):
- Radiation: UV light, X-rays, gamma rays.
- Pollutants: Air pollution, industrial chemicals.
- Drugs: e.g., paracetamol overdose (OD).
- Cigarette smoke: Rich in FRs.
- Inflammation: Chronic inflammation ↑ FRs.
⭐ The hydroxyl radical, often formed via the Fenton reaction, is considered the most reactive and damaging oxygen-derived free radical.
FR Damage - Radicals on Rampage
Free radicals wreak havoc on cellular components:
- Lipid Peroxidation: Polyunsaturated fatty acids (PUFAs) in membranes targeted.
- Chain reaction: initiation, propagation, termination.
- Markers: Malondialdehyde (MDA), 4-hydroxynonenal (4-HNE).
- Damage: ↑ Membrane permeability, ↓ fluidity, cell lysis.
- Protein Oxidation: Amino acid residues (Cys, Met) & protein backbone attacked.
- Markers: Protein carbonyls, 3-nitrotyrosine.
- Damage: Enzyme inactivation, protein aggregation & misfolding.
- DNA Damage: Bases (esp. Guanine) & deoxyribose sugar affected.
- Marker: $8-OHdG$ (8-hydroxy-2'-deoxyguanosine).
- Damage: Mutations, DNA strand breaks, carcinogenesis.
⭐ 8-OHdG is a critical biomarker for oxidative DNA damage, frequently elevated in various cancers and degenerative diseases, making it a common exam topic related to free radical injury assessment and antioxidant efficacy studies.
Antioxidant Defenses - Body's Superheroes
- Enzymatic Systems: Neutralize ROS. 📌 Mnemonic: Can Super Glue Protect? (Catalase, SOD, GPx)
- Superoxide Dismutase (SOD): Converts $O_2^{\cdot-}$ to $H_2O_2$. (Cu-Zn, Mn, EC types).
- Catalase (Peroxisomes): Degrades $H_2O_2$: $2H_2O_2 \rightarrow 2H_2O + O_2$.
- Glutathione Peroxidase (GPx-Se): Selenium-dependent. Reduces $H_2O_2$/lipid peroxides: $H_2O_2 + 2GSH \rightarrow GSSG + 2H_2O$.
- Glutathione Reductase: Regenerates GSH from GSSG (uses NADPH).
- Non-Enzymatic Scavengers:
- Glutathione (GSH): (\gamma)-Glu-Cys-Gly. Major intracellular antioxidant.
- Vitamin E ((\alpha)-tocopherol): Lipid-soluble; protects membranes. Chain-breaking.
- Vitamin C (Ascorbic acid): Water-soluble; regenerates Vit E; scavenges $O_2^{\cdot-}, OH^{\cdot}$.
- Uric Acid: Plasma antioxidant (purine metabolism).
- Bilirubin: Antioxidant (heme breakdown).
- Others: Carotenoids, flavonoids, melatonin.
⭐ Glutathione (GSH) depletion ↑ oxidative stress susceptibility (e.g., Parkinson's, liver damage).
Oxidative Stress & Disease - Health Under Siege
- Definition: Imbalance: ↑Reactive Oxygen/Nitrogen Species (ROS/RNS) vs. ↓Antioxidant capacity, causing cellular damage.
- Key Damage Markers:
- Lipids: Malondialdehyde (MDA).
- Proteins: Carbonyls.
- DNA: 8-hydroxy-2'-deoxyguanosine (8-OHdG).
- Associated Diseases:
- Atherosclerosis (LDL oxidation).
- Neurodegeneration (Alzheimer's, Parkinson's).
- Cancer (mutagenesis).
- Ischemia-reperfusion injury.
- Diabetes, chronic inflammation, aging.
⭐ 8-OHdG is a key biomarker for oxidative DNA damage, often elevated in various cancers and chronic diseases associated with oxidative stress.
High‑Yield Points - ⚡ Biggest Takeaways
- Key Reactive Oxygen Species (ROS): superoxide (O₂⁻•), hydroxyl radical (•OH), hydrogen peroxide (H₂O₂).
- Fenton reaction (Fe²⁺ + H₂O₂) generates highly reactive hydroxyl radicals (•OH).
- Superoxide Dismutase (SOD) (Mn-mitochondrial, Cu/Zn-cytosolic) converts O₂⁻• to H₂O₂.
- H₂O₂ is neutralized by Catalase and Selenium-dependent Glutathione Peroxidase.
- Glutathione (GSH), a key antioxidant, is regenerated by Glutathione Reductase (uses NADPH).
- Important antioxidant vitamins: Vitamin E (lipid-soluble), Vitamin C, and β-carotene.
- Lipid peroxidation damages membranes; marker: Malondialdehyde (MDA).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

