Carbohydrate Metabolism

On this page
🔋 The Cellular Energy Economy: Carbohydrate Command Center
Carbohydrate metabolism orchestrates how your body extracts, stores, and deploys energy-a process so fundamental that its disruption underlies conditions from diabetes to glycogen storage diseases. You'll master the enzymatic control points that regulate glucose flux, develop pattern recognition skills to distinguish metabolic disorders clinically, and learn evidence-based management strategies that connect biochemical pathways to bedside decisions. This lesson builds your ability to think systematically about energy homeostasis across organ systems, transforming abstract pathways into diagnostic and therapeutic tools you'll use daily in practice.
📌 Remember: GGGPFS - Glycolysis, Gluconeogenesis, Glycogenesis, Pentose phosphate, Fructose metabolism, Specialized sugars. These six pathways form the complete carbohydrate metabolic network, each serving distinct physiological roles during fed and fasted states.
The metabolic flexibility of carbohydrate pathways enables cells to respond to varying energy demands and substrate availability. During the fed state, insulin promotes anabolic pathways with glycogen synthesis increasing 300-fold and fatty acid synthesis rising 500%. Conversely, during fasting, glucagon and cortisol activate catabolic pathways, with gluconeogenesis contributing up to 60% of glucose production after 12 hours of fasting.
| Metabolic State | Primary Pathway | Key Hormone | Glucose Source | Duration | Clinical Significance |
|---|---|---|---|---|---|
| Fed State | Glycolysis | Insulin | Dietary glucose | 0-4 hours | Promotes storage, prevents hyperglycemia |
| Post-absorptive | Glycogenolysis | Glucagon | Liver glycogen | 4-12 hours | Maintains glucose for brain |
| Early Fasting | Gluconeogenesis | Cortisol | Amino acids | 12-24 hours | Preserves muscle glycogen |
| Prolonged Fasting | Ketogenesis | Growth hormone | Fatty acids | >24 hours | Glucose-sparing for brain |
| Starvation | Protein catabolism | Multiple | Muscle protein | >72 hours | Emergency glucose production |
- Glucose Transport Systems
- GLUT1: Brain and RBCs (Km = 1-2 mM, always saturated)
- GLUT2: Liver and pancreas (Km = 15-20 mM, glucose sensor)
- GLUT4: Muscle and adipose (insulin-dependent, 10-fold increase)
- Basal state: 5% of GLUT4 at membrane
- Insulin stimulation: 50% translocation within 15 minutes
- Exercise activation: Independent of insulin, additive effect
💡 Master This: Understanding GLUT kinetics predicts tissue glucose uptake patterns. GLUT1's low Km ensures constant brain glucose supply, while GLUT2's high Km allows liver to act as glucose buffer - taking up excess glucose when high, releasing when low.
The integration of carbohydrate metabolism with other metabolic pathways creates a sophisticated energy management system. Acetyl-CoA serves as the central metabolic hub, connecting carbohydrate oxidation to fatty acid synthesis (when abundant) or ketone production (when scarce). The glucose-fatty acid cycle (Randle cycle) ensures metabolic flexibility, with fatty acid oxidation inhibiting glycolysis by 70% through citrate accumulation and acetyl-CoA feedback.
⭐ Clinical Pearl: Metabolic syndrome disrupts normal carbohydrate handling through insulin resistance, affecting 25% of adults globally. Key markers include fasting glucose >100 mg/dL, HbA1c >5.7%, and 2-hour glucose tolerance >140 mg/dL, representing progressive loss of glucose homeostasis.
Understanding these foundational principles sets the stage for exploring the intricate regulatory mechanisms that fine-tune each pathway's activity in response to physiological demands.
🔋 The Cellular Energy Economy: Carbohydrate Command Center
⚙️ Metabolic Machinery: The Enzymatic Control Network
📌 Remember: RAPID regulation - Receptor binding, Allosteric changes, Phosphorylation cascades, Induction of enzymes, Degradation control. This sequence represents the temporal hierarchy of metabolic control from seconds to hours.
Key Regulatory Enzymes and Their Control Mechanisms:
-
Glycolysis Control Points
- Hexokinase: Product inhibition by glucose-6-phosphate (Ki = 0.1 mM)
- Phosphofructokinase-1: Master regulator with 6 allosteric sites
- Inhibitors: ATP (Ki = 0.5 mM), citrate (Ki = 0.3 mM)
- Activators: AMP (Ka = 0.1 mM), fructose-2,6-bisphosphate (Ka = 0.01 mM)
- Pyruvate kinase: Feed-forward activation by fructose-1,6-bisphosphate
-
Gluconeogenesis Control Points
- Pyruvate carboxylase: Activated by acetyl-CoA (Ka = 0.1 mM)
- PEPCK: Rate-limiting enzyme, induced 10-fold by glucagon
- Fructose-1,6-bisphosphatase: Inhibited by AMP and fructose-2,6-bisphosphate
- Reciprocal regulation with PFK-1 prevents futile cycling
- Energy cost of futile cycle: 1 ATP per glucose molecule
⭐ Clinical Pearl: The fructose-2,6-bisphosphate system acts as the master switch between glycolysis and gluconeogenesis. Insulin dephosphorylates PFK-2/F-2,6-BPase, increasing F-2,6-BP levels 5-fold and promoting glycolysis. Glucagon has the opposite effect, favoring gluconeogenesis.
| Enzyme | Pathway | Allosteric Activators | Allosteric Inhibitors | Covalent Modification | Clinical Significance |
|---|---|---|---|---|---|
| PFK-1 | Glycolysis | AMP, F-2,6-BP | ATP, Citrate | None | Metabolic flexibility |
| F-1,6-BPase | Gluconeogenesis | None | AMP, F-2,6-BP | None | Prevents futile cycling |
| Pyruvate kinase | Glycolysis | F-1,6-BP | ATP, Alanine | Phosphorylation (-) | Feed-forward control |
| PEPCK | Gluconeogenesis | None | None | Induction (+) | Glucose production |
| Acetyl-CoA carboxylase | Fatty acid synthesis | Citrate | Palmitoyl-CoA | Phosphorylation (-) | Metabolic switching |
💡 Master This: The glucose-fatty acid cycle (Randle cycle) creates metabolic flexibility through substrate competition. When fatty acids are oxidized, acetyl-CoA and citrate accumulate, inhibiting PFK-1 and pyruvate dehydrogenase. This mechanism explains why ketogenic diets reduce glucose utilization by 40-60%.
Tissue-Specific Metabolic Specialization:
-
Brain Metabolism
- Glucose consumption: 120-140 g/day (20% of total body glucose)
- Ketone adaptation: 50-70% glucose replacement during starvation
- Blood-brain barrier: GLUT1 transporters ensure constant glucose supply
-
Muscle Metabolism
- Type I fibers: Oxidative, high GLUT4 density, fatigue-resistant
- Type II fibers: Glycolytic, rapid glucose uptake, high power output
- Exercise adaptation: GLUT4 content increases 2-3 fold with training
-
Liver Metabolism
- Glucose buffer: Takes up 30% of portal glucose during fed state
- Gluconeogenic capacity: 150-200 g glucose/day during fasting
- Glycogen storage: 100-120 g (8-10% of liver weight)
⭐ Clinical Pearl: Dawn phenomenon affects 75% of type 1 diabetics, causing 50-100 mg/dL glucose rise between 4-8 AM. This results from growth hormone and cortisol surges promoting hepatic glucose production while insulin sensitivity decreases by 25%.
These regulatory mechanisms create the foundation for understanding how metabolic pathways integrate to maintain glucose homeostasis, setting the stage for examining specific pattern recognition frameworks in clinical practice.
⚙️ Metabolic Machinery: The Enzymatic Control Network
🎯 Clinical Pattern Recognition: The Metabolic Detective Framework
📌 Remember: FAST-HG pattern recognition - Fasting vs fed state, Age of onset, Symptom timing, Tissue affected, Hypoglycemia vs hyperglycemia, Genetic vs acquired. This framework systematically approaches any carbohydrate metabolism disorder.
Hypoglycemia Pattern Recognition Framework:
-
Timing-Based Classification
- Reactive hypoglycemia: Symptoms 2-4 hours post-meal
- Glucose nadir: <50 mg/dL at 2-3 hours
- Common causes: Dumping syndrome, early diabetes
- Key feature: Normal fasting glucose
- Fasting hypoglycemia: Symptoms after >8 hours fasting
- Glucose levels: <45 mg/dL (men), <40 mg/dL (women)
- Serious causes: Insulinoma, adrenal insufficiency
- Critical threshold: <30 mg/dL causes neuroglycopenia
- Reactive hypoglycemia: Symptoms 2-4 hours post-meal
-
Whipple's Triad Recognition
- Symptoms consistent with hypoglycemia (100% required)
- Low glucose during symptomatic episodes (<55 mg/dL)
- Symptom relief with glucose administration (within 10-15 minutes)
- Sensitivity: 100% for pathological hypoglycemia
- Specificity: 85% (false positives in reactive cases)
⭐ Clinical Pearl: The insulin:glucose ratio during hypoglycemia distinguishes causes. Ratio >0.3 suggests inappropriate insulin secretion (insulinoma, sulfonylurea use), while ratio <0.3 indicates appropriate insulin suppression (adrenal insufficiency, liver disease).
| Clinical Presentation | Glucose Pattern | Key Laboratory Findings | Diagnostic Threshold | Treatment Response |
|---|---|---|---|---|
| Insulinoma | Fasting hypoglycemia | Insulin >3 μU/mL, C-peptide >0.6 ng/mL | 72-hour fast positive | Diazoxide responsive |
| Factitious insulin | Variable timing | Insulin >3 μU/mL, C-peptide <0.6 ng/mL | Insulin:C-peptide >1 | Octreotide resistant |
| Sulfonylurea abuse | Post-meal timing | Insulin >3 μU/mL, C-peptide >0.6 ng/mL | Positive drug screen | Octreotide responsive |
| Adrenal insufficiency | Fasting predominant | Cortisol <3 μg/dL, ACTH >100 pg/mL | Cosyntropin test | Steroid replacement |
| Liver disease | Progressive pattern | ALT >3x normal, albumin <3 g/dL | Child-Pugh score | Glucose infusion |
-
Diabetic Ketoacidosis (DKA) Triad
- Hyperglycemia: Usually >250 mg/dL (range: 200-800 mg/dL)
- Ketosis: β-hydroxybutyrate >3 mM or urine ketones ≥2+
- Acidosis: pH <7.3 and bicarbonate <15 mEq/L
- Anion gap: >12 mEq/L (typically 15-25 mEq/L)
- Mortality: <1% with appropriate treatment
-
Hyperosmolar Hyperglycemic State (HHS)
- Severe hyperglycemia: >600 mg/dL (often >1000 mg/dL)
- Hyperosmolality: >320 mOsm/kg (calculated or measured)
- Minimal ketosis: β-hydroxybutyrate <3 mM
- Mortality: 5-15% (higher than DKA)
- Neurological symptoms: Focal deficits in 25% of cases
💡 Master This: The effective osmolality calculation distinguishes HHS from DKA: 2(Na + K) + glucose/18. Values >320 mOsm/kg indicate HHS, while <320 mOsm/kg with ketosis suggests DKA. Mixed presentations occur in 30% of cases.
Glycogen Storage Disease (GSD) Patterns:
-
Hepatic GSDs (Types I, III, VI, IX)
- Hepatomegaly: >5 cm below costal margin
- Fasting hypoglycemia: <40 mg/dL after 4-6 hours
- Growth retardation: Height <3rd percentile
- Laboratory pattern: ↑Lactate, ↑uric acid, ↑triglycerides
-
Myopathic GSDs (Types II, V, VII)
- Exercise intolerance: Fatigue within 5-10 minutes
- "Second wind" phenomenon: Improvement after 10-15 minutes
- CK elevation: 5-50x normal (baseline 200-2000 U/L)
- Myoglobinuria: Risk with intense exercise
⭐ Clinical Pearl: The ischemic forearm test differentiates myopathic GSDs. Normal lactate rise (3-5x baseline) excludes muscle glycogenoses. Flat lactate response suggests Type V (McArdle) or Type VII (Tarui) disease.
These pattern recognition frameworks provide the foundation for systematic differential diagnosis, leading to targeted investigations and evidence-based treatment approaches.
🎯 Clinical Pattern Recognition: The Metabolic Detective Framework
🔬 Systematic Discrimination: The Metabolic Differential Matrix
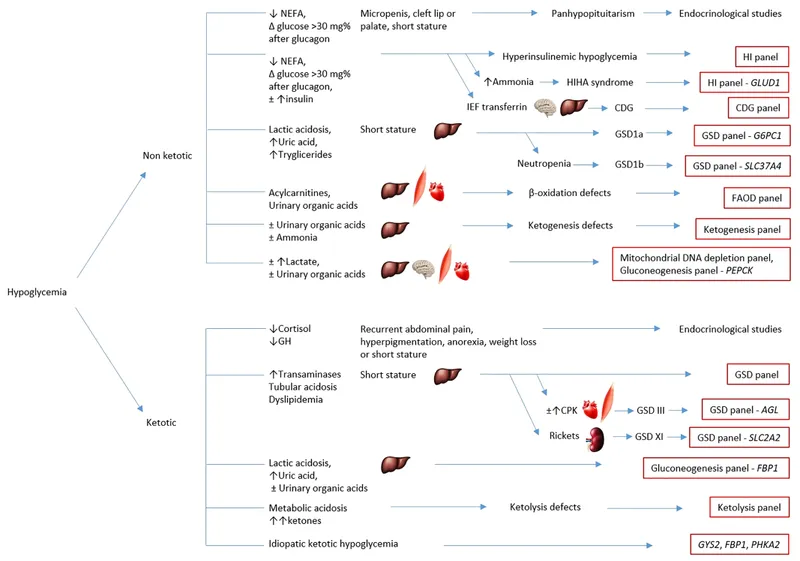
📌 Remember: DIVIDE systematic approach - Duration of symptoms, Inheritance pattern, Vital organ involvement, Insulin/glucose dynamics, Dietary triggers, Enzyme deficiencies. This framework systematically discriminates between carbohydrate metabolism disorders.
Hypoglycemia Differential Matrix:
| Disorder | Onset Age | Fasting Duration | Insulin Level | C-peptide | Key Discriminator | Diagnostic Test |
|---|---|---|---|---|---|---|
| Insulinoma | 40-60 years | 12-72 hours | >6 μU/mL | >0.6 ng/mL | Inappropriate insulin | 72-hour fast |
| Nesidioblastosis | <1 year | 2-4 hours | >10 μU/mL | >1.0 ng/mL | Persistent hyperinsulinism | Genetic testing |
| KATP mutations | Neonatal | 1-2 hours | >15 μU/mL | >1.5 ng/mL | Diazoxide unresponsive | Molecular analysis |
| Adrenal insufficiency | Variable | 8-12 hours | <2 μU/mL | <0.3 ng/mL | Low cortisol response | Cosyntropin test |
| Growth hormone deficiency | 2-5 years | 6-8 hours | <3 μU/mL | <0.4 ng/mL | Poor growth velocity | IGF-1 levels |
| Glycogen storage disease | Infancy | 4-6 hours | <2 μU/mL | <0.3 ng/mL | Hepatomegaly + lactate | Enzyme assay |
- Normal ratio: <1.0 (equimolar secretion)
- Exogenous insulin: >1.0 (C-peptide suppressed)
- Sulfonylurea: <1.0 (both elevated proportionally)
- Insulin autoantibodies: Variable (interferes with assays)
⭐ Clinical Pearl: Proinsulin levels provide additional discrimination. Insulinomas secrete >22% proinsulin (normal <20%), while factitious insulin administration shows <5% proinsulin due to processed insulin injection.
Diabetes Mellitus Subtype Discrimination:
-
Type 1 vs Type 2 Diabetes Distinguishing Features
- Age of onset: <30 years (85% Type 1) vs >40 years (90% Type 2)
- BMI at diagnosis: <25 kg/m² (70% Type 1) vs >30 kg/m² (80% Type 2)
- C-peptide levels: <0.5 ng/mL (Type 1) vs >1.0 ng/mL (Type 2)
- Autoantibody presence: >90% positive (Type 1) vs <5% positive (Type 2)
-
MODY (Maturity-Onset Diabetes of Young) Characteristics
- Family history: 3 generations affected (autosomal dominant)
- Age of onset: <25 years in ≥2 family members
- Non-insulin dependent: >2 years without insulin
- Specific mutations: HNF1A (50%), GCK (30%), HNF4A (10%)
Glycogen Storage Disease Discrimination:
| GSD Type | Enzyme Defect | Primary Organ | Hypoglycemia | Exercise Intolerance | Key Laboratory Finding |
|---|---|---|---|---|---|
| Type I (von Gierke) | Glucose-6-phosphatase | Liver | Severe (<40 mg/dL) | No | Lactate >4 mM |
| Type II (Pompe) | α-1,4-glucosidase | Heart/Muscle | No | Yes | CK >1000 U/L |
| Type III (Cori) | Debranching enzyme | Liver/Muscle | Moderate (40-60 mg/dL) | Mild | Normal lactate |
| Type V (McArdle) | Muscle phosphorylase | Muscle | No | Severe | Flat lactate response |
| Type VI (Hers) | Liver phosphorylase | Liver | Mild (50-70 mg/dL) | No | Mild hepatomegaly |
Ketosis Differential Analysis:
-
Diabetic Ketoacidosis vs Starvation Ketosis
- Glucose levels: >250 mg/dL (DKA) vs <100 mg/dL (starvation)
- Ketone levels: >5 mM (DKA) vs 1-3 mM (starvation)
- pH status: <7.3 (DKA) vs >7.35 (starvation)
- Insulin levels: Low but detectable (DKA) vs Very low (starvation)
-
Alcoholic Ketoacidosis Characteristics
- Glucose levels: Normal to low (80-150 mg/dL)
- Ketone pattern: β-hydroxybutyrate predominant (>10:1 ratio)
- Anion gap: 15-25 mEq/L (similar to DKA)
- Response: Rapid improvement with glucose + thiamine
⭐ Clinical Pearl: The β-hydroxybutyrate:acetoacetate ratio distinguishes ketosis types. DKA shows 3:1 ratio, starvation shows 2:1 ratio, while alcoholic ketoacidosis shows >10:1 ratio due to altered NADH/NAD+ balance.
These systematic discrimination tools provide the analytical framework for evidence-based treatment decisions and optimal patient outcomes.
🔬 Systematic Discrimination: The Metabolic Differential Matrix
⚖️ Treatment Algorithms: Evidence-Based Metabolic Management
📌 Remember: TREAT systematic approach - Time-sensitive recognition, Rapid stabilization, Evidence-based protocols, Adjusted monitoring, Targeted endpoints. This framework ensures optimal outcomes in metabolic emergencies.
Diabetic Ketoacidosis (DKA) Management Protocol:
-
Fluid Management Priorities
- Initial assessment: Hemodynamic stability and degree of dehydration
- Severe dehydration: Normal saline 1-2 L in first hour
- Maintenance fluids: 250-500 mL/hour based on cardiac status
- Electrolyte monitoring: Every 2-4 hours during acute phase
- Sodium correction: Expected rise 2-4 mEq/L per 100 mg/dL glucose drop
- Potassium replacement: Start when K+ <5.0 mEq/L and urine output adequate
-
Insulin Protocol Specifications
- Loading dose: 0.1 U/kg IV bolus (optional, not routinely recommended)
- Continuous infusion: 0.1 U/kg/hour (typical range: 5-10 units/hour)
- Glucose target: Decrease 50-75 mg/dL/hour (not faster)
- Ketone clearance: Primary endpoint (glucose normalization secondary)
⭐ Clinical Pearl: Bicarbonate therapy is contraindicated unless pH <6.9. Studies show no benefit for pH 6.9-7.1 and potential harm including cerebral edema risk (0.5-1% incidence) and paradoxical CNS acidosis.
| DKA Severity | pH Range | Bicarbonate | Anion Gap | Mental Status | Monitoring Frequency |
|---|---|---|---|---|---|
| Mild | 7.25-7.30 | 15-18 mEq/L | 10-12 mEq/L | Alert | Every 4 hours |
| Moderate | 7.00-7.24 | 10-15 mEq/L | >12 mEq/L | Alert/drowsy | Every 2 hours |
| Severe | <7.00 | <10 mEq/L | >12 mEq/L | Stupor/coma | Every 1 hour |
-
Conscious Patient Treatment
- Mild symptoms (glucose 54-70 mg/dL): 15 g oral glucose
- Moderate symptoms (glucose 40-54 mg/dL): 20 g oral glucose
- Severe symptoms (glucose <40 mg/dL): 25 g oral glucose or IV access
- Recheck timing: 15 minutes after treatment, retreat if needed
-
Unconscious Patient Protocol
- IV access available: 25 g dextrose (50 mL D50) IV push
- No IV access: Glucagon 1 mg IM/SQ (0.5 mg if <20 kg)
- Expected response: Consciousness within 10-15 minutes
- Follow-up: Complex carbohydrates once alert and able to swallow
💡 Master This: Glucagon effectiveness depends on glycogen stores. Ineffective in starvation, chronic hypoglycemia, or alcohol-induced hypoglycemia. Success rate: 85% in well-nourished patients, <50% in malnourished patients.
Glycogen Storage Disease Management:
-
Type I GSD (von Gierke) Protocol
- Dietary management: Continuous glucose supply via cornstarch
- Cornstarch dosing: 1.6-2.5 g/kg every 3-4 hours (including nighttime)
- Target glucose: >70 mg/dL at all times
- Monitoring: Home glucose every 4 hours, HbA1c <7%
- Complications: Annual screening for hepatic adenomas (>80% develop)
-
Type II GSD (Pompe) Treatment
- Enzyme replacement: Alglucosidase alfa 20 mg/kg every 2 weeks
- Response monitoring: 6-minute walk test, pulmonary function
- Cardiac assessment: Echo every 6 months (infantile form)
- Prognosis: Infantile form - median survival 8.7 months (untreated)
⭐ Clinical Pearl: Liver transplantation in Type I GSD corrects glucose-6-phosphatase deficiency but doesn't address kidney disease. Success rate: >90% survival, but progressive nephropathy continues in 30% of patients.
Diabetes Management Targets:
| Patient Population | HbA1c Target | Fasting Glucose | Postprandial Glucose | Blood Pressure | LDL Cholesterol |
|---|---|---|---|---|---|
| Healthy adults | <7.0% | 80-130 mg/dL | <180 mg/dL | <140/90 mmHg | <100 mg/dL |
| Elderly/comorbid | <8.0% | 90-150 mg/dL | <200 mg/dL | <150/90 mmHg | <100 mg/dL |
| Pregnancy | <6.0% | <95 mg/dL | <140 mg/dL | <135/85 mmHg | <100 mg/dL |
| Pediatric | <7.5% | 90-130 mg/dL | <180 mg/dL | <90th percentile | <100 mg/dL |
⚖️ Treatment Algorithms: Evidence-Based Metabolic Management
🌐 Multi-System Integration: The Metabolic Connectome
📌 Remember: CONNECT integration framework - Cardiovascular coupling, Organ crosstalk, Neural networks, Nutritional sensing, Endocrine coordination, Circadian timing, Tissue specialization. This system-level view reveals metabolic integration complexity.
Liver-Muscle-Adipose Metabolic Triangle:
-
Fed State Coordination
- Liver: Glucose uptake 30%, glycogen synthesis ↑300%, lipogenesis ↑500%
- Muscle: Glucose uptake 80% (via GLUT4 translocation), protein synthesis ↑200%
- Adipose: Glucose uptake 15%, lipogenesis ↑400%, lipolysis ↓90%
- Integration signal: Insulin coordinates anabolic programs across all tissues
-
Fasted State Coordination
- Liver: Glucose production 2-3 mg/kg/min, ketogenesis ↑1000%
- Muscle: Protein catabolism ↑150%, amino acid release for gluconeogenesis
- Adipose: Lipolysis ↑500%, free fatty acid release >1 mM
- Integration signals: Glucagon, cortisol, growth hormone coordinate catabolic programs
⭐ Clinical Pearl: The hepatic glucose sensor (GLUT2) enables the liver to function as a glucose buffer. During hyperglycemia, liver glucose uptake increases proportionally without saturation, while during hypoglycemia, glucose production maintains brain glucose supply at >4 mM.
| Metabolic State | Liver Function | Muscle Contribution | Adipose Role | Brain Adaptation | Integration Hormone |
|---|---|---|---|---|---|
| Fed (0-4h) | Glucose uptake | Glucose storage | Lipid storage | Glucose utilization | Insulin |
| Post-absorptive (4-12h) | Glucose production | Glycogen breakdown | Minimal lipolysis | Glucose dependent | Glucagon |
| Fasting (12-24h) | Gluconeogenesis | Protein catabolism | Active lipolysis | Glucose sparing | Cortisol |
| Starvation (>24h) | Ketogenesis | Muscle preservation | Maximal lipolysis | Ketone adaptation | Growth hormone |
-
Incretin System Integration
- GLP-1 secretion: L-cells respond to glucose, amino acids, fatty acids
- Pancreatic effects: β-cell insulin secretion ↑300%, α-cell glucagon ↓50%
- Gastric effects: Emptying delayed by 40-60%, satiety increased
- CNS effects: Hypothalamic appetite suppression, reward pathway modulation
-
Neural Glucose Sensing
- Hypothalamic glucose sensors: Detect 5-10% glucose changes
- Sympathetic activation: Hepatic glucose production ↑200% within minutes
- Parasympathetic response: Insulin secretion priming, digestive preparation
- Brainstem integration: Glucose counter-regulation during hypoglycemia
Circadian Metabolic Integration:
-
Clock Gene Regulation
- CLOCK/BMAL1: Master regulators of metabolic gene expression
- Tissue-specific rhythms: Liver (60% genes rhythmic), muscle (40%), adipose (30%)
- Metabolic oscillations: Glucose tolerance varies 40% throughout 24-hour cycle
- Insulin sensitivity: Peak at 8 AM, nadir at 8 PM (25% difference)
-
Chronotherapy Applications
- Metformin timing: Evening dosing improves dawn phenomenon by 30%
- Insulin regimens: Basal-bolus patterns match physiological rhythms
- Meal timing: Early eating improves glucose tolerance by 20%
- Exercise timing: Post-meal activity reduces glucose excursions by 50%
💡 Master This: Metabolic flexibility represents the ability to switch between glucose and fatty acid oxidation based on substrate availability. Healthy individuals achieve 50% substrate switching within 3-6 hours, while metabolic syndrome patients require 12-24 hours.
Exercise-Induced Metabolic Integration:
-
Acute Exercise Responses
- Muscle glucose uptake: ↑10-50 fold during intense exercise
- Hepatic glucose production: Matches muscle uptake (normally 2-3 mg/kg/min)
- Catecholamine surge: Epinephrine ↑20-fold, norepinephrine ↑10-fold
- Lactate production: ↑15-20 fold with anaerobic threshold
-
Training Adaptations
- GLUT4 content: ↑2-3 fold in trained muscle
- Mitochondrial density: ↑50-100% with endurance training
- Insulin sensitivity: ↑40-50% lasting 24-48 hours post-exercise
- Glycogen storage: ↑20-40% in trained individuals
⭐ Clinical Pearl: Exercise-induced glucose uptake occurs through insulin-independent mechanisms via AMPK activation and calcium signaling. This explains why exercise remains effective for glucose control even in severe insulin resistance.
Stress Response Integration:
-
Acute Stress (Fight-or-Flight)
- Cortisol release: ↑5-10 fold within 30 minutes
- Glucose production: ↑200-300% via gluconeogenesis
- Insulin resistance: ↑50% in peripheral tissues
- Duration: Returns to baseline within 2-6 hours
-
Chronic Stress Effects
- HPA axis dysregulation: Persistent cortisol elevation
- Metabolic consequences: Insulin resistance, central obesity, dyslipidemia
- Glucose intolerance: ↑40% risk of diabetes development
- Therapeutic targets: Stress management, cortisol modulators
These multi-system integration patterns reveal how carbohydrate metabolism functions as part of a sophisticated whole-body regulatory network, setting the foundation for advanced clinical mastery tools.
🌐 Multi-System Integration: The Metabolic Connectome
🎯 Clinical Mastery Arsenal: Rapid-Fire Metabolic Command
📌 Remember: MASTER clinical arsenal - Memory tools, Algorithms, Scoring systems, Thresholds, Emergency protocols, Rapid recognition. These tools transform complex metabolism into actionable clinical decisions.
Essential Clinical Thresholds:
-
Glucose Critical Values
- Severe hypoglycemia: <40 mg/dL (immediate intervention required)
- Moderate hypoglycemia: 40-54 mg/dL (symptomatic, treat promptly)
- Mild hypoglycemia: 54-70 mg/dL (asymptomatic, monitor closely)
- Normal range: 70-100 mg/dL (fasting), <140 mg/dL (2-hour post-meal)
- Diabetes threshold: ≥126 mg/dL (fasting), ≥200 mg/dL (random + symptoms)
- HHS threshold: >600 mg/dL (often >1000 mg/dL)
-
Ketosis Severity Markers
- Trace ketosis: 0.5-1.0 mM β-hydroxybutyrate
- Mild ketosis: 1.0-3.0 mM (nutritional ketosis range)
- Moderate ketosis: 3.0-5.0 mM (concerning, investigate)
- Severe ketosis: >5.0 mM (DKA range, immediate treatment)
- Life-threatening: >15 mM (ICU management required)
⭐ Clinical Pearl: Point-of-care β-hydroxybutyrate testing provides results in 10 seconds with 98% correlation to laboratory values. Cost-effective for DKA monitoring and reduces hospital length of stay by average 12 hours.
Rapid Diagnostic Algorithms:
| Clinical Scenario | First Test | Critical Threshold | Immediate Action | Next Step |
|---|---|---|---|---|
| Unconscious patient | Glucose | <50 mg/dL | D50 25g IV | Investigate cause |
| Vomiting + diabetes | Ketones | >3.0 mM | IV fluids + insulin | DKA protocol |
| Severe dehydration | Osmolality | >320 mOsm/kg | Aggressive fluids | HHS management |
| Neonatal seizures | Glucose | <40 mg/dL | D10 2-4 mL/kg | Hyperinsulinism workup |
| Exercise intolerance | CK + lactate | CK >1000 U/L | Avoid intense exercise | GSD evaluation |
-
Hypoglycemia Treatment
- Conscious adult: 15-20g oral glucose (repeat in 15 minutes if needed)
- Unconscious adult: 25g dextrose (50 mL D50) IV push
- Pediatric: 0.5-1 g/kg dextrose (2-4 mL/kg D25)
- Glucagon: 1 mg IM/SQ (adult), 0.5 mg (child <20 kg)
- Octreotide: 50-100 μg SQ q8h (hyperinsulinism)
-
DKA Insulin Protocol
- Loading dose: 0.1 U/kg IV (optional)
- Continuous infusion: 0.1 U/kg/hour (typical 5-10 U/hour)
- Pediatric rate: 0.05-0.1 U/kg/hour (lower starting dose)
- Glucose target: Decrease 50-75 mg/dL/hour
- Transition: Overlap SQ insulin 1-2 hours before stopping IV
💡 Master This: Two-bag system for DKA management allows independent adjustment of insulin and dextrose rates. Bag 1: Normal saline + electrolytes. Bag 2: D10 + electrolytes. Switch between bags based on glucose levels while maintaining constant insulin.
Pattern Recognition Mnemonics:
📌 Hypoglycemia Causes: EXPLAIN - Exogenous insulin, Xtra exercise, Poor intake, Liver disease, Adrenal insufficiency, Insulinoma, Nesidioblastosis
📌 DKA Precipitants: MISSED - Myocardial infarction, Infection, Stroke, Surgery/stress, Endocrine disorders, Drugs (steroids, diuretics)
📌 GSD Hepatic Types: "1-3-6-9 Liver Alive" - Types I, III, VI, IX cause hepatomegaly and hypoglycemia
📌 GSD Muscle Types: "2-5-7 Muscles Heaven" - Types II, V, VII cause exercise intolerance and muscle symptoms
Monitoring Parameters:
-
DKA Resolution Criteria (ALL must be met)
- Glucose: <200 mg/dL AND
- Bicarbonate: ≥15 mEq/L AND
- pH: >7.3 AND
- Anion gap: ≤12 mEq/L AND
- Mental status: Normal
-
Hypoglycemia Workup Priorities
- During episode: Glucose, insulin, C-peptide, β-hydroxybutyrate
- Additional tests: Cortisol, growth hormone, drug screen
- Timing critical: Samples before glucose administration
- 72-hour fast: Gold standard for fasting hypoglycemia
⭐ Clinical Pearl: Whipple's triad must be documented during spontaneous episodes. Provoked hypoglycemia (exercise, fasting) has different diagnostic significance than spontaneous episodes and may not require extensive workup.
This clinical mastery arsenal provides the rapid-access tools essential for confident management of carbohydrate metabolism disorders across emergency, inpatient, and outpatient settings.
🎯 Clinical Mastery Arsenal: Rapid-Fire Metabolic Command
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app
























