AA Catabolism Intro - Nitrogen's Journey Begins
- Surplus amino acids (AAs) are degraded; no dedicated storage.
- Core task: Channel nitrogen from AAs for safe disposal or reuse.
- Removal of the $\alpha$-amino group is the obligatory first step.
- Yields: carbon skeleton (for energy/biosynthesis) & a nitrogenous part.
- Nitrogenous part primarily forms ammonia ($NH_3$) or is transferred to specific carriers.
- $NH_3$ is highly toxic; requires detoxification (e.g., urea cycle in liver).

⭐ The $\alpha$-amino group of most amino acids is ultimately transferred to $\alpha$-ketoglutarate to form glutamate, which acts as a central collector of amino groups.
Transamination - Amino Group Relay
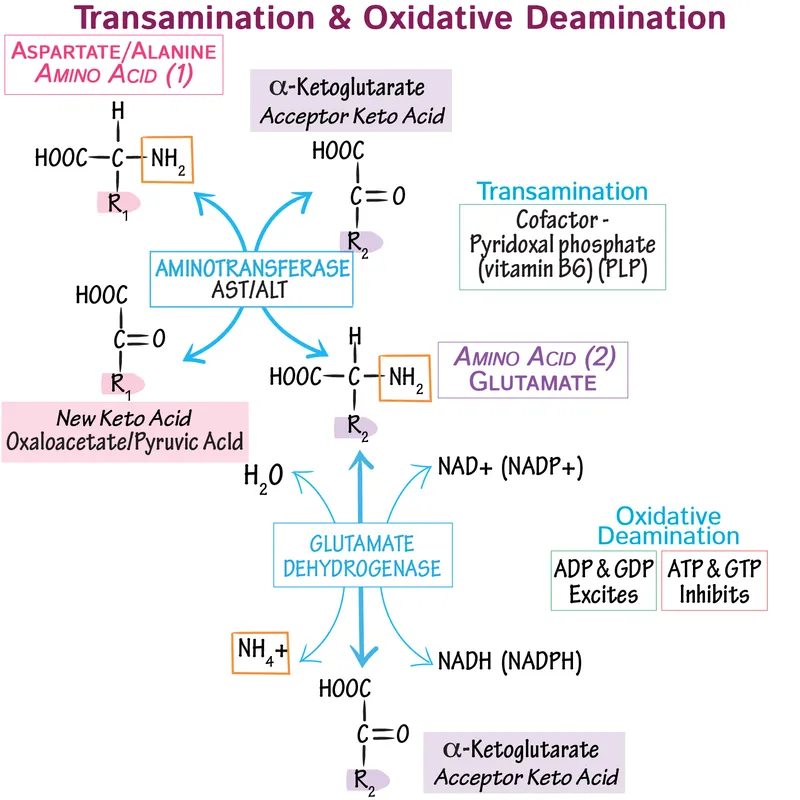
- Reversible transfer of an amino ($\text{-NH}_2$) group from one L-amino acid to an α-keto acid, yielding a new α-keto acid and a new L-amino acid.
- Enzymes: Aminotransferases (also called Transaminases).
- Coenzyme: Pyridoxal Phosphate (PLP), an active Vitamin B6 derivative, is crucial.
- Site: Primarily liver cytosol & mitochondria; also other tissues.
- Significance:
- Key for interconversion of amino acids (e.g., non-essential AA synthesis).
- Collects amino groups from diverse amino acids onto L-glutamate, the central amino group carrier.
- 📌 Exceptions (do not transaminate): Lysine, Threonine, Proline, Hydroxyproline. (Mnemonic: "Lying Three Pros Hide")
⭐ Serum ALT (Alanine Aminotransferase) & AST (Aspartate Aminotransferase) are key liver function tests (LFTs); ALT is more liver-specific.
coenzyme role in enzymatic transamination)
Oxidative Deamination - Glutamate's Big Release
- Primary mechanism for $NH_4^+$ liberation from amino acids, mainly in liver & kidney mitochondria.
- Key Enzyme: Glutamate Dehydrogenase (GDH).
- Catalyzes: Glutamate + $NAD(P)^+$ + $H_2O$ $\rightleftharpoons$ $\alpha$-Ketoglutarate + $NH_4^+$ + $NAD(P)H$ + $H^+$.
- Reversible; direction depends on cellular energy state & substrate levels.
- Regulation (Allosteric):
- Activators (signal low energy): ADP, GDP.
- Inhibitors (signal high energy): ATP, GTP.
- Significance:
- Provides $NH_4^+$ for urea synthesis.
- $\alpha$-Ketoglutarate enters TCA cycle for energy or gluconeogenesis.
- 📌 Mnemonic: GDH: Glutamate Delivers Hydrogen (and Ammonia!).
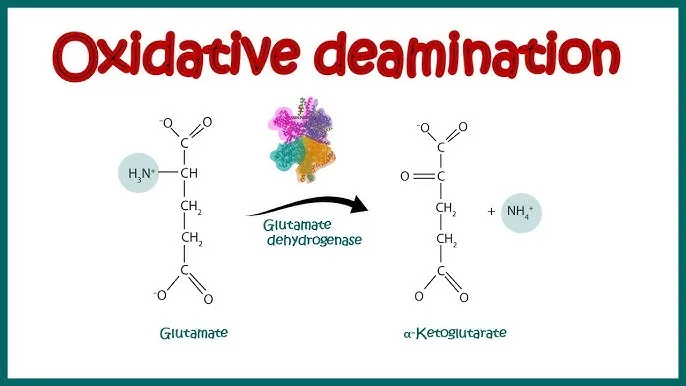
⭐ GDH is unique among dehydrogenases as it can utilize both $NAD^+$ and $NADP^+$ as coenzymes, linking amino acid catabolism to anabolic pathways when $NADP^+$ is used (e.g., fatty acid synthesis).
Non-Oxidative Deamination - Direct Ammonia Drop
- Direct $NH_3$ removal from select AAs; no oxidation. Mostly PLP-dependent.
- 📌 AAs: Serine, Threonine, Cysteine, Homocysteine, Histidine.
- Serine/Threonine Dehydratase (PLP):
- Serine $\rightarrow$ Pyruvate + $NH_4^+$
- Threonine $\rightarrow$ $\alpha$-ketobutyrate + $NH_4^+$
- Desulfhydrases (PLP): For sulfur AAs (Cys, Hcy).
- Cysteine $\rightarrow$ Pyruvate + $NH_4^+$ + $H_2S$
- Histidase (No PLP):
- Histidine $\rightarrow$ Urocanate + $NH_4^+$
- Deficiency: Histidinemia.
⭐ Histidase (Histidine $\rightarrow$ Urocanate + $NH_4^+$) deficiency causes Histidinemia, a key inborn error of metabolism.
Transdeamination & Clinical Links - Pathway Power Duo
- Transdeamination: Major pathway for amino acid catabolism. Sequential action of:
- Aminotransferases (e.g., ALT, AST; cofactor: PLP - Pyridoxal Phosphate)
- Glutamate Dehydrogenase (GDH) in liver mitochondria.
- Overall Process: Amino group from AA $\rightarrow$ Glutamate $\rightarrow$ $NH_4^+$ (for urea cycle).
- Glutamate + $NAD(P)^+$ $\xrightarrow{GDH}$ $\alpha$-Ketoglutarate + $NH_4^+$ + $NAD(P)H$.
- Clinical Significance:
- Liver damage: ↑ serum ALT (SGPT) & AST (SGOT).
- Hepatic encephalopathy: From ↑ blood $NH_3$.
⭐ ALT is more liver-specific than AST. AST also found in heart, muscle. (📌 ALT: L for Liver).
High‑Yield Points - ⚡ Biggest Takeaways
- Transamination by aminotransferases (e.g., ALT, AST) requires PLP (B6), collecting amino groups on glutamate.
- ALT is more liver-specific; AST is also in heart and muscle.
- Oxidative deamination of glutamate by mitochondrial Glutamate Dehydrogenase (GDH) releases NH₄⁺ for the urea cycle.
- GDH uses NAD⁺/NADP⁺ and is allosterically regulated: inhibited by ATP/GTP, activated by ADP/GDP.
- Non-oxidative deamination occurs for serine and threonine.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more