Pain Management

On this page
🎯 The Pain Command Center: Mastering Clinical Pain Management
Pain is both a vital protective signal and one of medicine's most complex clinical challenges, demanding that you master not just its neurophysiology but the systematic approach to decode what each patient's pain reveals. You'll build expertise from the neural pathways that transmit nociceptive signals through the clinical pattern recognition that distinguishes somatic from neuropathic presentations, then advance to evidence-based treatment algorithms that match mechanism to intervention. This lesson equips you with a command center approach: integrating anatomy, diagnostic precision, and therapeutic strategy to transform pain assessment from subjective complaint into actionable clinical intelligence.
📌 Remember: PQRST Assessment Framework - Provocation/Palliation, Quality, Radiation, Severity (0-10 scale), Timing. This systematic approach captures 85% of diagnostically relevant pain characteristics and guides evidence-based treatment selection.
The foundation of effective pain management rests on understanding pain as a complex neurobiological process involving multiple pathways, receptors, and modulation systems. Modern pain medicine recognizes pain not merely as a symptom but as a distinct pathophysiological entity requiring targeted, mechanism-based interventions.
- Nociceptive Pain Characteristics
- Mechanical threshold: >15 mN force activation
- Thermal threshold: >43°C heat, <15°C cold
- Chemical mediators: Prostaglandins (PGE2), bradykinin, substance P
- Response time: 0.5-2 seconds for Aδ fibers
- Delayed response: 2-30 seconds for C fibers
- Neuropathic Pain Features
- Prevalence: 7-10% of general population
- Diagnostic accuracy: >90% with DN4 questionnaire score ≥4
- Treatment response: 30-50% achieve >50% pain reduction
- First-line agents: Gabapentin 300-3600 mg/day
- Alternative: Pregabalin 150-600 mg/day
⭐ Clinical Pearl: Pain scores >7/10 correlate with 60% increased risk of chronic pain development and require aggressive early intervention. Patients reporting pain >6/10 for >3 months show 40% reduced response to standard analgesics.
| Pain Type | Onset Pattern | Duration | Response to Opioids | Key Features | Treatment Success |
|---|---|---|---|---|---|
| Acute Nociceptive | Immediate | <3 months | Excellent (80-90%) | Proportional to tissue damage | 85-95% |
| Chronic Nociceptive | Gradual | >3 months | Moderate (40-60%) | Persistent inflammation | 60-75% |
| Neuropathic | Variable | Often chronic | Poor (20-40%) | Burning, shooting, electric | 30-50% |
| Mixed | Complex | Variable | Variable (30-70%) | Combined features | 45-65% |
| Psychogenic | Inconsistent | Variable | Minimal (10-20%) | Disproportionate presentation | 25-40% |
The clinical assessment framework integrates quantitative pain measurement with qualitative descriptors to guide mechanism-based treatment selection. Effective pain management begins with precise characterization of pain patterns, triggers, and associated symptoms.
Connect this foundational understanding through neurophysiological mechanisms to understand how pain signals transform from peripheral stimuli into conscious perception.
🎯 The Pain Command Center: Mastering Clinical Pain Management
⚡ Neural Highway Architecture: Pain Transmission Mastery
📌 Remember: ABCD Fiber Classification - Aα (proprioception, 80-120 m/s), Aβ (touch, 35-75 m/s), Aδ (fast pain, 5-30 m/s), C fibers (slow pain, 0.5-2 m/s). Fiber diameter and myelination determine conduction velocity and pain quality.
The peripheral nociceptive system demonstrates remarkable specificity in detecting and encoding harmful stimuli. Understanding nociceptor subtypes and their activation thresholds guides targeted peripheral interventions.
- Nociceptor Subtypes and Activation
- Mechanical nociceptors: Threshold >15 mN, respond to tissue deformation
- Thermal nociceptors: Heat >43°C, cold <15°C activation
- Polymodal nociceptors: 70% of C fibers, multiple stimulus types
- Sensitization threshold: pH <6.5, inflammatory mediators
- Recovery time: 15-45 minutes post-stimulus
- Spinal Cord Processing Zones
- Lamina I: Nociceptive-specific neurons, 15% of dorsal horn
- Lamina II (Substantia Gelatinosa): Modulation center, 25% interneurons
- Lamina V: Wide dynamic range neurons, 40% convergence
- Gate control mechanism: Aβ fiber input inhibits nociception
- Temporal summation: >0.3 Hz C fiber stimulation

⭐ Clinical Pearl: Local anesthetic concentration determines fiber blockade selectivity. 0.25% bupivacaine blocks C fibers preferentially, while 0.5% concentration blocks Aδ fibers, and >0.75% affects motor Aα fibers.
| Ascending Tract | Origin | Destination | Function | Clinical Significance | Fiber Composition |
|---|---|---|---|---|---|
| Spinothalamic | Lamina I, V | VPL Thalamus | Discriminative pain | Stroke localization | 90% crossed |
| Spinoreticular | Lamina VII, VIII | Reticular formation | Arousal, autonomic | Consciousness level | Bilateral |
| Spinomesencephalic | Lamina I | Periaqueductal gray | Descending control | Opioid analgesia | 80% crossed |
| Spinohypothalamic | Lamina I | Hypothalamus | Neuroendocrine | Stress response | Bilateral |
| Spinocervical | Lamina IV | Cervical nuclei | Touch modulation | Gate control | Ipsilateral |
The descending pain control system provides powerful endogenous analgesia through multiple neurotransmitter pathways. This system represents the primary target for many analgesic medications and explains individual variations in pain sensitivity.

- Descending Control Centers
- Periaqueductal Gray (PAG): Opioid receptor density >500 fmol/mg
- Rostral Ventromedial Medulla (RVM): Serotonin and norepinephrine
- Locus Coeruleus: Noradrenergic modulation, α2 receptors
- Stimulation produces 70-90% pain reduction
- Duration: 30-120 minutes depending on stimulus intensity
📌 Remember: SEND Neurotransmitters - Serotonin (5-HT), Endorphins (opioids), Norepinephrine (NE), Dopamine. These descending modulators explain antidepressant analgesic effects and combination therapy rationale.
Connect this transmission architecture through clinical pain states to understand how pathway dysfunction creates distinct pain syndromes requiring targeted interventions.
⚡ Neural Highway Architecture: Pain Transmission Mastery
🔧 Clinical Pain Decoder: Pattern Recognition Mastery
📌 Remember: SOCRATES Pain Assessment - Site, Onset, Character, Radiation, Associations, Time course, Exacerbating/relieving factors, Severity. This framework captures >95% of diagnostically relevant information for pain syndrome classification.
The acute pain recognition framework focuses on identifying underlying pathophysiology and predicting treatment response patterns. Acute pain characteristics directly correlate with tissue damage extent and inflammatory response magnitude.
- Acute Pain Recognition Patterns
- Somatic Pain: Well-localized, sharp/aching quality
- Intensity: Proportional to tissue damage
- Duration: <6 weeks typical resolution
- Response: 80-90% improvement with NSAIDs + opioids
- Visceral Pain: Poorly localized, cramping/pressure quality
- Referred patterns: Predictable dermatomal distribution
- Autonomic signs: >70% have nausea, diaphoresis
- Response: 60-75% improvement with antispasmodics
- Somatic Pain: Well-localized, sharp/aching quality
- Chronic Pain Transformation Markers
- Risk factors: Pain >7/10 for >2 weeks
- Psychological factors: Catastrophizing score >30
- Sleep disruption: <6 hours nightly for >1 month
- Chronification risk: 40-60% with multiple factors
- Prevention window: <12 weeks for intervention
⭐ Clinical Pearl: Pain that awakens patients from sleep suggests organic pathology in >85% of cases and requires urgent evaluation. Functional pain rarely disrupts sleep architecture, while inflammatory and neuropathic pain characteristically cause sleep fragmentation.
| Pain Syndrome | Key Descriptors | Associated Features | Diagnostic Tests | First-Line Treatment | Success Rate |
|---|---|---|---|---|---|
| Acute Nociceptive | Sharp, throbbing | Swelling, warmth | Clinical exam | NSAIDs + opioids | 85-95% |
| Neuropathic | Burning, electric | Allodynia, hyperalgesia | EMG, nerve conduction | Gabapentin/pregabalin | 30-50% |
| Fibromyalgia | Widespread aching | Fatigue, sleep issues | Tender points (11/18) | Duloxetine/milnacipran | 40-60% |
| Complex Regional | Burning, throbbing | Color/temperature changes | Bone scan, thermography | Sympathetic blocks | 50-70% |
| Myofascial | Deep aching | Trigger points, referred | Physical exam | Trigger point injection | 70-85% |
- Neuropathic Pain Diagnostic Criteria
- DN4 Questionnaire: Score ≥4 indicates neuropathic pain (90% sensitivity)
- Sensory abnormalities: Allodynia in >60%, hyperalgesia in >80%
- Pain descriptors: "Burning" (75%), "electric" (65%), "shooting" (55%)
- Temporal patterns: Continuous background pain + paroxysmal episodes
- Distribution: Follows anatomical nerve territories
- Chronic Pain Syndrome Markers
- Central sensitization: Widespread hyperalgesia beyond injury site
- Psychological comorbidity: Depression in >50%, anxiety in >40%
- Functional impairment: Work disability in >30% of chronic pain patients
- Sleep disruption: >80% report poor sleep quality
- Cognitive effects: "Brain fog" in >60% of patients
💡 Master This: Pain that responds poorly to opioids but improves with anticonvulsants suggests neuropathic mechanisms. This pattern recognition guides early treatment selection and prevents ineffective opioid escalation.
📌 Remember: LANSS Scale - Lancinating pain, Allodynia, Neurological deficits, Sensory changes, Skin changes. Scores ≥12 indicate neuropathic pain with >85% accuracy and guide anticonvulsant therapy selection.
Connect these recognition patterns through systematic diagnostic approaches to understand how clinical assessment guides evidence-based treatment selection and outcome prediction.
🔧 Clinical Pain Decoder: Pattern Recognition Mastery
🔍 Diagnostic Precision Matrix: Systematic Pain Analysis
📌 Remember: VINDICATE Differential Framework - Vascular, Infectious, Neoplastic, Degenerative, Iatrogenic, Congenital, Autoimmune, Traumatic, Endocrine. This systematic approach ensures comprehensive evaluation of pain etiologies.
The diagnostic precision matrix integrates multiple assessment domains to create comprehensive pain profiles that guide mechanism-based treatment selection. Each diagnostic category requires specific evaluation approaches and therapeutic considerations.
- Primary Assessment Domains
- Temporal Characteristics: Acute (<3 months), subacute (3-6 months), chronic (>6 months)
- Anatomical Distribution: Localized, regional, widespread patterns
- Mechanism Classification: Nociceptive (70%), neuropathic (20%), mixed (10%)
- Response predictors: Mechanism determines treatment success rates
- Diagnostic accuracy: >90% with systematic assessment
- Advanced Diagnostic Criteria
- Quantitative Sensory Testing: Thermal thresholds, vibration detection
- Functional Assessment: Pain interference scores, disability indices
- Psychological Screening: Depression (PHQ-9), anxiety (GAD-7), catastrophizing
- Comorbidity impact: >50% reduction in treatment response with untreated depression
- Screening accuracy: >85% with validated instruments
⭐ Clinical Pearl: Pain that varies significantly with weather changes suggests inflammatory mechanisms in >75% of cases, while pain unaffected by activity or position suggests neuropathic or central mechanisms in >80% of presentations.
| Diagnostic Category | Key Features | Confirmatory Tests | Treatment Response | Prognosis | Monitoring Parameters |
|---|---|---|---|---|---|
| Inflammatory | Morning stiffness >1hr | ESR >30, CRP >10 | Excellent to NSAIDs | Good with treatment | Inflammatory markers |
| Neuropathic | Allodynia, hyperalgesia | EMG abnormalities | Poor to opioids | Variable, often chronic | Sensory testing |
| Ischemic | Exercise-related | Vascular studies | Good to revascularization | Depends on intervention | Perfusion studies |
| Mechanical | Position-dependent | Imaging abnormalities | Good to correction | Excellent if correctable | Functional assessment |
| Psychogenic | Inconsistent patterns | Normal objective tests | Variable response | Requires psychological care | Psychological metrics |
- Specialized Diagnostic Approaches
- Neuropathic Pain Assessment: Nerve conduction studies, skin biopsy
- Small fiber neuropathy: >50% have normal EMG
- Diagnostic yield: Skin biopsy >80% sensitivity
- Central Sensitization Evaluation: Widespread hyperalgesia, temporal summation
- Clinical markers: Pain beyond tissue damage, multiple tender points
- Prevalence: >30% of chronic pain patients show central features
- Neuropathic Pain Assessment: Nerve conduction studies, skin biopsy
- Treatment Response Patterns
- Opioid responsiveness: Nociceptive >80%, neuropathic <40%
- Anti-inflammatory response: Inflammatory >90%, neuropathic <20%
- Anticonvulsant efficacy: Neuropathic >60%, nociceptive <30%
- Response timeline: 2-4 weeks for anticonvulsants, 1-3 days for NSAIDs
- Dose-response: Linear for NSAIDs, plateau effect for anticonvulsants
💡 Master This: Diagnostic accuracy improves from 60% with clinical assessment alone to >90% when combining clinical evaluation, validated questionnaires, and treatment response patterns. Systematic assessment prevents misclassification and guides optimal therapy selection.
📌 Remember: PAINS Red Flags - Progressive neurological deficits, Age >50 with new onset, Immune compromise, Night pain disrupting sleep, Systemic symptoms (fever, weight loss). These features require urgent evaluation and imaging studies.
Connect this diagnostic precision through evidence-based treatment algorithms to understand how systematic assessment guides optimal therapeutic intervention selection and outcome optimization.
🔍 Diagnostic Precision Matrix: Systematic Pain Analysis
⚖️ Therapeutic Command Center: Evidence-Based Treatment Algorithms
📌 Remember: STEPS Treatment Hierarchy - Simple analgesics first, Targeted mechanism therapy, Escalation protocols, Procedural interventions, Specialist referral. This systematic approach achieves >80% treatment success with minimal adverse effects.
The evidence-based treatment framework integrates multiple therapeutic modalities based on pain mechanism, severity, and patient-specific factors. Understanding treatment hierarchies and combination strategies optimizes outcomes while minimizing risks.
- First-Line Treatment Strategies
- Acetaminophen: 1000mg q6h (maximum 4g/day)
- Efficacy: 30-40% pain reduction in mild-moderate pain
- Onset: 30-60 minutes, duration 4-6 hours
- Safety: Hepatotoxicity risk at >4g/day or with alcohol
- NSAIDs: Ibuprofen 400-800mg q8h, naproxen 500mg q12h
- Anti-inflammatory effect: >50% reduction in inflammatory markers
- Cardiovascular risk: 1.5-2x increased risk with long-term use
- GI protection: PPI required with >65 years or risk factors
- Acetaminophen: 1000mg q6h (maximum 4g/day)
- Mechanism-Specific Interventions
- Neuropathic Pain: Gabapentin 300-3600mg/day, pregabalin 150-600mg/day
- Response rate: 50-60% achieve >50% pain reduction
- Time to effect: 1-2 weeks for initial response, 4-8 weeks for maximum benefit
- Inflammatory Pain: Corticosteroids, disease-modifying agents
- Steroid efficacy: >70% improvement in inflammatory conditions
- Duration: 2-4 weeks for oral steroids, 6-12 weeks for injections
- Neuropathic Pain: Gabapentin 300-3600mg/day, pregabalin 150-600mg/day
⭐ Clinical Pearl: Combination therapy achieves >60% pain reduction in 75% of patients compared to 45% with monotherapy. Synergistic effects occur when combining different mechanism targets, particularly opioids with anticonvulsants for neuropathic pain.
| Treatment Category | Mechanism | Efficacy Rate | Time to Effect | Duration | Major Limitations |
|---|---|---|---|---|---|
| Acetaminophen | Central COX inhibition | 40-50% | 30-60 min | 4-6 hours | Hepatotoxicity risk |
| NSAIDs | Peripheral COX inhibition | 60-70% | 15-30 min | 6-12 hours | GI/CV/renal toxicity |
| Opioids | μ-receptor agonism | 70-80% | 15-30 min | 3-6 hours | Dependence, tolerance |
| Anticonvulsants | Sodium/calcium channels | 50-60% | 1-2 weeks | Continuous | Sedation, dizziness |
| Antidepressants | Monoamine reuptake | 40-60% | 2-4 weeks | Continuous | Anticholinergic effects |
- Multimodal Combination Strategies
- Acute Pain Protocol: Acetaminophen + NSAID + opioid PRN
- Opioid reduction: 30-50% decreased requirement
- Side effect reduction: 40% less nausea, sedation
- Chronic Pain Management: Anticonvulsant + antidepressant + topical agents
- Synergistic effects: >70% achieve meaningful improvement
- Quality of life: 50-60% improvement in functional scores
- Acute Pain Protocol: Acetaminophen + NSAID + opioid PRN
- Interventional Pain Procedures
- Nerve blocks: 70-90% success rate for appropriate candidates
- Epidural injections: 60-80% improvement lasting 3-6 months
- Radiofrequency ablation: >80% pain reduction for 6-12 months
- Patient selection: Critical for success rates
- Diagnostic blocks: >80% pain relief predicts procedure success
💡 Master This: Treatment success correlates with mechanism matching - neuropathic pain requires anticonvulsants/antidepressants, inflammatory pain responds to NSAIDs/steroids, and nociceptive pain improves with opioids. Mismatched therapy explains treatment failures and guides algorithm adjustments.
📌 Remember: SMART Goals for Pain Management - Specific targets (pain scores), Measurable outcomes (functional improvement), Achievable expectations (30-50% reduction realistic), Relevant to patient priorities, Time-bound assessments (2-4 week intervals).
Connect these evidence-based algorithms through advanced integration strategies to understand how complex pain conditions require sophisticated multimodal approaches and specialist coordination.
⚖️ Therapeutic Command Center: Evidence-Based Treatment Algorithms
🔗 Advanced Integration Hub: Complex Pain Mastery
📌 Remember: BIOPSYCHOSOCIAL Model - Biological mechanisms, Psychological factors, Social determinants all influence pain perception and treatment response. Addressing all domains achieves >70% success rates compared to <40% with single-domain approaches.
The advanced integration framework combines multiple therapeutic modalities with precision timing and dosing to achieve synergistic effects while minimizing adverse outcomes. Understanding complex interactions enables optimization of difficult-to-treat pain conditions.
- Complex Pain Syndrome Management
- Central Sensitization Syndromes: Fibromyalgia, chronic widespread pain
- Prevalence: 2-4% of population, >80% female predominance
- Treatment approach: Duloxetine 60-120mg/day + pregabalin 300-450mg/day
- Success metrics: >30% pain reduction in 60% of patients
- Chronic Regional Pain Syndrome (CRPS): Complex sympathetic dysfunction
- Early intervention: <6 months critical for optimal outcomes
- Treatment protocol: Sympathetic blocks + physical therapy + medications
- Recovery rates: >80% with early aggressive treatment, <40% if delayed
- Central Sensitization Syndromes: Fibromyalgia, chronic widespread pain
- Cutting-Edge Treatment Modalities
- Neuromodulation: Spinal cord stimulation, peripheral nerve stimulation
- Patient selection: Failed conservative therapy, >50% pain reduction with trial
- Success rates: 70-80% achieve meaningful improvement
- Technology advances: Closed-loop systems, high-frequency stimulation
- Regenerative Medicine: Platelet-rich plasma, stem cell therapy
- Evidence level: Emerging data, 30-50% improvement in select conditions
- Applications: Osteoarthritis, tendinopathies, neuropathic pain
- Neuromodulation: Spinal cord stimulation, peripheral nerve stimulation
⭐ Clinical Pearl: Patients with >3 failed medication trials benefit from interdisciplinary pain programs with 65% achieving clinically meaningful improvement compared to 25% with continued medication adjustments alone.
| Integration Strategy | Components | Success Rate | Duration | Cost-Effectiveness | Patient Selection |
|---|---|---|---|---|---|
| Interdisciplinary Care | Medical + PT + Psychology | 65-75% | 3-6 months | High long-term value | Chronic complex pain |
| Neuromodulation | Device + medication optimization | 70-80% | Permanent implant | Moderate-high | Failed conservative care |
| Interventional Series | Multiple procedures + rehab | 60-70% | 6-12 months | Moderate | Specific anatomical targets |
| Intensive Rehabilitation | PT + OT + psychology | 55-65% | 2-4 months | High value | Functional impairment |
| Pharmacogenomics | Genetic testing + targeted meds | 50-60% | Ongoing | Emerging evidence | Multiple drug failures |
- Personalized Medicine Applications
- Pharmacogenomic Testing: CYP2D6, CYP3A4 polymorphisms affect opioid metabolism
- Clinical impact: 2-10x variation in drug levels based on genetics
- Cost-effectiveness: >$3000 savings per patient with multiple drug trials
- Biomarker-Guided Therapy: Inflammatory markers, neuropeptide levels
- Predictive accuracy: >80% for treatment response in select populations
- Applications: TNF-α levels predict anti-inflammatory response
- Pharmacogenomic Testing: CYP2D6, CYP3A4 polymorphisms affect opioid metabolism
- Technology Integration
- Digital Health Platforms: Mobile apps, wearable devices, telemedicine
- Engagement rates: >70% with gamification and feedback systems
- Outcome improvement: 20-30% additional benefit with digital support
- Artificial Intelligence: Pattern recognition, treatment optimization
- Diagnostic accuracy: >90% for complex pain syndrome classification
- Treatment prediction: >80% accuracy for medication response
- Digital Health Platforms: Mobile apps, wearable devices, telemedicine
💡 Master This: Complex pain conditions require systems thinking - addressing biological mechanisms, psychological factors, social determinants, and functional goals simultaneously. Single-modality approaches fail because pain involves multiple interconnected systems requiring coordinated intervention.
📌 Remember: INTEGRATION Principles - Interdisciplinary teams, Neuromodulation options, Technology utilization, Evidence-based protocols, Genetic considerations, Rehabilitation focus, Advanced procedures, Tailored approaches, Innovative therapies, Outcome optimization, Network coordination.
Connect this advanced integration understanding through practical mastery tools to develop rapid clinical decision-making frameworks and essential reference systems for optimal pain management outcomes.
🔗 Advanced Integration Hub: Complex Pain Mastery
🎯 Clinical Mastery Arsenal: Rapid Pain Management Command
📌 Remember: RAPID Assessment Protocol - Recognize pain pattern, Assess severity/function, Plan mechanism-based therapy, Implement multimodal approach, Determine response and adjust. This 5-step framework guides systematic pain management in <5 minutes.
Essential Clinical Arsenal:
- Pain Emergency Protocols
- Severe Acute Pain (>8/10): Morphine 0.1mg/kg IV q15min PRN or fentanyl 1-2mcg/kg IV
- Breakthrough Cancer Pain: Immediate-release opioid 10-20% of daily dose
- Neuropathic Crisis: Gabapentin 300mg TID + topical lidocaine 5%
- Response timeline: 15-30 minutes for IV opioids, 2-4 hours for oral agents
- Monitoring: Respiratory rate >12/min, sedation score <3
- Rapid Diagnostic Differentials
- Red Flag Pain: Progressive neurological deficits, fever, night sweats
- Neuropathic Indicators: Burning, electric, allodynia, hyperalgesia
- Inflammatory Markers: Morning stiffness >1 hour, joint swelling
- Diagnostic accuracy: >90% with systematic assessment
- Urgent referral: Cauda equina, compartment syndrome, necrotizing fasciitis
⭐ Clinical Pearl: The "Rule of 10s" - Pain >7/10 for >10 days with <10% improvement requires treatment escalation or specialist referral. This simple rule prevents prolonged suffering and identifies treatment-resistant cases early.
| Clinical Scenario | First-Line Treatment | Backup Option | Red Flags | Expected Timeline | Success Criteria |
|---|---|---|---|---|---|
| Acute Injury Pain | Ibuprofen 600mg + ice | Add tramadol 50mg | Deformity, neurovascular compromise | 24-48 hours | Pain <4/10, functional |
| Postoperative Pain | Multimodal protocol | PCA opioids | Severe uncontrolled pain | 2-5 days | Mobilization possible |
| Neuropathic Pain | Gabapentin 300mg TID | Pregabalin 75mg BID | Progressive weakness | 1-2 weeks | 30% pain reduction |
| Cancer Pain | Morphine per WHO ladder | Fentanyl patch | Rapid escalation needs | 24-72 hours | Comfort achieved |
| Chronic Pain Flare | Increase baseline meds | Short steroid course | New neurological signs | 3-7 days | Return to baseline |
- Essential Drug Dosing Quick Reference
- Morphine Equivalents: Morphine 10mg = Oxycodone 6.7mg = Hydromorphone 1.5mg
- Anticonvulsant Dosing: Gabapentin start 300mg TID, max 3600mg/day
- NSAID Maximums: Ibuprofen 2400mg/day, naproxen 1000mg/day
- Conversion accuracy: ±10% for equianalgesic dosing
- Titration schedule: 25-50% increases every 3-7 days
- Critical Monitoring Parameters
- Opioid Safety: Respiratory rate >12, sedation score, naloxone availability
- NSAID Monitoring: Creatinine, blood pressure, GI symptoms
- Anticonvulsant Watch: Dizziness, peripheral edema, mood changes
- Frequency: Daily for acute, weekly for chronic adjustments
- Documentation: Pain scores, functional status, adverse effects
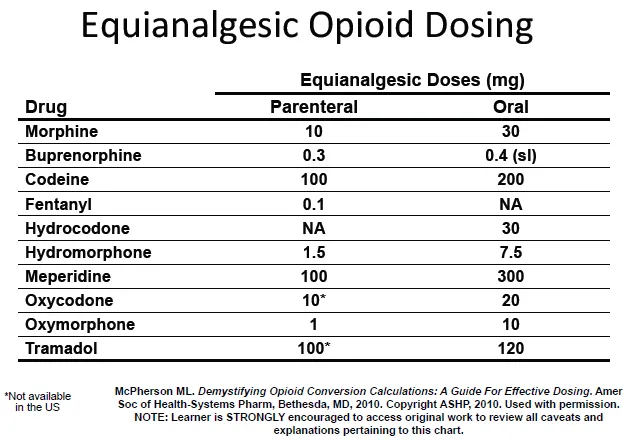
💡 Master This: Successful pain management requires pattern recognition + protocol adherence + outcome monitoring. Master these three domains, and you possess the foundation for excellent pain care in any clinical setting.
📌 Remember: SUCCESS Metrics - Severity reduction >30%, Understandable treatment plan, Compliance achievable, Comorbidities addressed, Expectations realistic, Side effects manageable, Specialist referral when needed. These criteria define optimal pain management outcomes.
🎯 Clinical Mastery Arsenal: Rapid Pain Management Command
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more
Have doubts about this lesson?
Ask Rezzy, your AI Study Mate, to explain anything you didn't understand
Everything you need for NEET-PG prep
Get full Oncourse access with lessons, practice questions, flashcards and AI study tools.
Scan to download app























